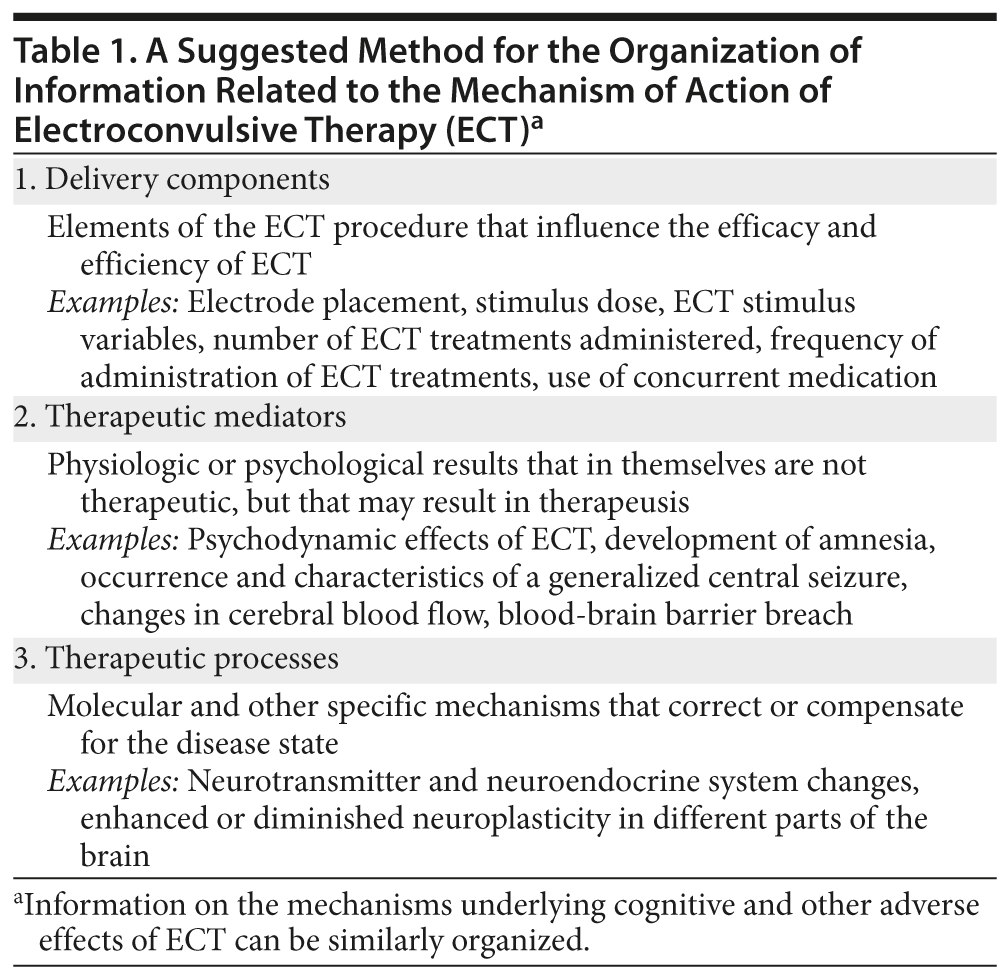
- Information on the mechanism of action of electroconvulsive therapy (ECT) can be organized under 3 headings: delivery components, therapeutic mediators, and therapeutic processes.
- Delivery components are elements of the ECT procedure that influence the response rate and the speed of response; examples are electrical dose, electrode placement, and number and frequency of ECT treatments administered.
- Therapeutic mediators are neurobiological changes that are not themselves therapeutic but that lead to therapeutic changes; an example is the central seizure.
- Therapeutic processes are the biological changes that may explain clinical improvement; an example is decreased neuroplasticity in the amygdala, which might diminish the emotional distress attached to stressful life events.
ABSTRACT
Much literature is available on the effects of electroconvulsive therapy (ECT) in the brain. Clinicians need to know how to organize this information when they explain hypotheses about the mechanism of action of ECT to patients and caregivers. One possibility is to classify the data under the headings of delivery components, therapeutic mediators, and therapeutic processes. Delivery components are elements of the ECT procedure that modulate the efficacy and efficiency of the treatment; examples are electrical dose, electrode placement, and number and frequency of treatments administered. Therapeutic mediators are physiologic or psychological changes that in themselves are not therapeutic but that lead to the suggested therapeutic changes; examples are the occurrence of the seizure and the occurrence of blood-brain barrier breach. Therapeutic processes are the actual biological changes that compensate for or correct the biological disturbances that underlie the psychiatric illness; examples are changes in the activity of certain neurotransmitter systems and increases or decreases in neuroplasticity in different parts of the brain. Organizing information in this manner can help explain both efficacy and adverse effects of ECT. Brief explanations are provided.
J Clin Psychiatry 2014;75(6):e548-e551 (doi:10.4088/JCP.14f09234)
© Copyright 2014 Physicians Postgraduate Press, Inc.
Clinical Problem
The previous article in this column1 presented an antidepressant-refractory, severely depressed patient for whom electroconvulsive therapy (ECT) had been suggested. The patient had asked why it was necessary for electricity to be passed through his brain. He wanted to know how ECT acts. The article1 explained why the question about the mechanism of action of ECT is a complex one and why it needs to be resolved into specific elements. The article also explained difficulties in the interpretation of research and academic concerns related to the generation of explanatory models. The present article deals with the problem of plenty; that is, how the large body of evidence on the subject may be organized so as to generate coherent explanations about the mechanism of action of ECT.
Much evidence is available on the electrophysiologic, neurochemical, neurotransmitter, neuroendocrine, histologic, and other changes that result with ECT, most or all of which have been offered as explanations for its mechanism of action. A considerable problem that one faces is to understand which of these changes are therapeutic and which are epiphenomena; which of the therapeutic changes are upstream and which are downstream; and how to fit electricity, seizure, premedication and comedication, and other elements into the model.
One possible way of organizing the information is to consider it under the headings of delivery components, therapeutic mediators, and therapeutic processes (Table 1).2
Delivery Components
Delivery components are the therapeutic elements of the ECT procedure that influence the rate and the extent to which a patient responds to ECT. Thus, the therapeutic elements determine the efficacy (response and remission rates) and the efficiency (speed of response to treatment) of ECT.
There are many clinically significant delivery components (Table 1). Bilateral ECT is generally associated with better chances of treatment response than unilateral ECT,3 possibly because of the use of higher electrical doses4 and because of better generalization of the central seizure.5 High-dose ECT is associated with higher response rates than low-dose ECT when electrode placement is unilateral,6-8 and when high-dose unilateral ECT is administered, response rates may equal those with bilateral ECT.7,9 High-dose ECT is also associated with faster clinical response, regardless of electrode placement.6 In this context, ECT dosage is generally reckoned in units of charge; that is, millicoulombs.4,10
The benefits of ECT cumulate across the ECT course, indicating that the number of ECT treatments administered is a necessary criterion for optimum improvement11; however, the rate at which these treatments are administered is also important because evidence suggests that more frequent ECT is associated with quicker antidepressant response when thrice- vs twice-weekly ECT are compared and with greater chances of response when thrice- vs once-weekly ECT are compared.12
Concurrent medication may improve treatment outcomes with ECT.13 Some14 but not all15 data suggest that ketamine anesthesia may improve ECT outcomes, perhaps through modulation of glutamatergic neurotransmission. Other delivery components may also have direct or indirect effects on clinical outcomes by influencing the ECT seizure or by direct effects on the brain; these range from aspects of premedication (eg, anticholinergic medication use) to aspects of the ECT procedure (eg, hyperventilation, to lower the seizure threshold). In fact, it is possible that even differences in the components (pulse amplitude, pulse width, pulse frequency, and stimulus duration) of the electrical stimulus may result in different neurobiological effects and hence in differences in clinical outcomes.16,17
Why are ECT delivery components important to an organization of research findings about the mechanism of action of ECT? The most important reason is that if variations in delivery components produce variations in treatment outcomes, then the neurobiological differences associated with these variations may be related to the neurobiological mechanisms responsible for the variations in outcomes. Some examples follow:
- Bilateral ECT is associated with greater seizure generalization than unilateral ECT.5 Given that the occurrence of a seizure is essential for therapeusis, this suggests that seizure mechanisms mediate the greater efficacy of bilateral ECT in depression.
- Relative to low-dose ECT, high-dose ECT is associated with greater suppression of neuroplasticity in the amygdala.18,19 This suggests that better suppression of negative affect related to noxious memories may explain why high-dose ECT is a more effective treatment.
Neurobiological variations associated with delivery components also provide other clues about ECT mechanisms. For example, at constant electrical dose, variations in the electrical components of the ECT stimulus result in variations in ictal, postictal, and interictal electroencephalographic (EEG) markers of ECT efficacy.16 As another example, ultrabrief-pulse ECT, which may be less effective than brief-pulse ECT,20 is associated with poorer induction of hippocampal neuroplasticity,17 strengthening the suggestion that neuroplasticity changes are at least in part responsible for antidepressant action.21-23
Therapeutic Mediators
Therapeutic mediators (Table 1) are physiologic or psychological effects of ECT that in themselves are not therapeutic but that may result in beneficial changes. Historically, a number of psychological hypotheses were proposed (but now discarded), including that ECT causes regression, fear, or a sense of punishment and that these effects psychodynamically result in absolution from guilt and, thereby, in attenuation of depression. Another discarded hypothesis is that ECT-induced amnesia attenuates the memory of the stressor and hence lessens depression.24,25
For over half a century, the occurrence of a generalized seizure has been recognized as the sine qua non for ECT-induced therapeutic benefits,26 and measures of seizure efficacy that have been proposed include seizure duration (presently discredited; see Andrade26), changes in seizure threshold,27 and patterns of EEG activity during, immediately after, and between ECT seizures.16,28,29 Neurobiological changes that the seizure induces have been well described, and these include neurohormonal and neurotransmitter changes,24,30-33 gene transcription,34 and neuroplasticity.35-37
Other treatment mediators include increases in cerebral blood flow and blood-brain barrier breach; there is, however, no convincing evidence in support of these as treatment mediators.38
Therapeutic Processes
Therapeutic processes are neurohormonal, neurotransmitter, neuroreceptor, second messenger system, genetic transcription, protein synthesis, neuroplasticity, and other specific changes that occur with ECT and that correct or compensate for the disease state. Therapeutic processes (with special reference to neuroplasticity) will be dealt with in greater detail in the next article in this series. To be strictly correct, strengthening or weakening of neuronal pathways, or increased or decreased structural neuroplasticity changes, are probably the only final common pathway therapeutic processes; however, given how little we understand about the brain in health and disease, it is reasonable to subsume all the mechanisms that lead to these ends as therapeutic processes.
Synthesis
Far too much information is available on the mechanism of action of ECT to be covered in a single article; readers may prefer to consult other reviews for additional information.23-25,30-33,39 For reasons explained in the previous article in this series,1 one cannot offer even a single hypothesis with certainty. Therefore, the present article does not seek to be either comprehensive or reductionistic. Rather, it seeks to provide a framework for putting evidence together and for understanding and communicating information about ECT. By way of example, a clinician may explain ECT as follows:
Delivery of a small but adequate dose of electricity to the brain results in the elicitation of seizure activity in the brain; higher electrical doses elicit a better quality of seizure. The seizure releases a large number of signaling chemicals that, in turn, produce changes on the surface and then within nerve and glial cells in target areas of the brain such as the hippocampus, amygdala, and prefrontal cortex. Genes in these cells are activated or suppressed, which in turn leads to increase or decrease in the number, activity, and connectivity of these cells. Increased number and connectivity of cells in the hippocampus and prefrontal cortex probably improve thinking and coping abilities, whereas decreased number and connectivity of cells in the amygdala reduce negative emotions attached to unpleasant memories; both effects can be expected to be therapeutic in depressed patients. The time course of recovery from depression seems to parallel the time course of occurrence of these cellular changes.
Detailed though such an explanation is, it is nevertheless a simplification and might need to be further simplified when educating laypersons about the mechanism of action of ECT. There is certainly a lot more to the mechanism of action of ECT, and certainly other explanations as well, as an earlier article indicated.1 However, the explanation provided is a reasonable hypothesis, at least for the beneficial action of ECT in depression3 and posttraumatic stress disorder.40
Parting Note
Adverse effects of ECT can also be explained in terms of delivery components, mediators, and processes (Table 1). A discussion
 Each month in his online column, Dr Andrade considers theoretical and practical ideas in clinical psychopharmacology with a view to update the knowledge and skills of medical practitioners who treat patients with psychiatric conditions.
Each month in his online column, Dr Andrade considers theoretical and practical ideas in clinical psychopharmacology with a view to update the knowledge and skills of medical practitioners who treat patients with psychiatric conditions.
Department of Clinical Psychopharmacology and Neurotoxicology, National Institute of Mental Health and Neurosciences, Bangalore, India ([email protected]).
Financial disclosure and more about Dr Andrade.on the subject, however, is out of the scope of the present article.
REFERENCES
1. Andrade C. A primer for the conceptualization of the mechanism of action of electroconvulsive therapy, 1: defining the question. J Clin Psychiatry. 2014;75(5):e410-e412.
2. Andrade C. The mechanism of action of ECT. In: Gangadhar BN, ed. Proceedings of the National Workshop on ECT: Priorities for Research and Practice in India. Bangalore, India: NIMHANS; 1992:107-130.
3. UK ECT Review Group. Efficacy and safety of electroconvulsive therapy in depressive disorders: a systematic review and meta-analysis. Lancet. 2003;361(9360):799-808. PubMed doi:10.1016/S0140-6736(03)12705-5
4. Sackeim HA, Decina P, Portnoy S, et al. Studies of dosage, seizure threshold, and seizure duration in ECT. Biol Psychiatry. 1987;22(3):249-268. PubMed doi:10.1016/0006-3223(87)90144-2
5. Abrams R. A hypothesis to explain divergent findings among studies comparing the efficacy of unilateral and bilateral ECT in depression. Convuls Ther. 1986;2(4):253-257. PubMed
6. Sackeim HA, Prudic J, Devanand DP, et al. Effects of stimulus intensity and electrode placement on the efficacy and cognitive effects of electroconvulsive therapy. N Engl J Med. 1993;328(12):839-846. PubMed doi:10.1056/NEJM199303253281204
7. Sackeim HA, Prudic J, Devanand DP, et al. A prospective, randomized, double-blind comparison of bilateral and right unilateral electroconvulsive therapy at different stimulus intensities. Arch Gen Psychiatry. 2000;57(5):425-434. PubMed doi:10.1001/archpsyc.57.5.425
8. McCall WV, Reboussin DM, Weiner RD, et al. Titrated moderately suprathreshold vs fixed high-dose right unilateral electroconvulsive therapy: acute antidepressant and cognitive effects. Arch Gen Psychiatry. 2000;57(5):438-444. PubMed doi:10.1001/archpsyc.57.5.438
9. McCall WV, Dunn A, Rosenquist PB, et al. Markedly suprathreshold right unilateral ECT versus minimally suprathreshold bilateral ECT: antidepressant and memory effects. J ECT. 2002;18(3):126-129. PubMed doi:10.1097/00124509-200209000-00003
10. Andrade C. Quantifying the ECT dose: the right unit remains elusive. J ECT. 2001;17(1):75. PubMed doi:10.1097/00124509-200103000-00017
11. Andrade C. Electroconvulsive therapy. In: Bhugra D, Ranjith G, Patel V, eds. Handbook of Psychiatry: A South Asian Perspective. New Delhi, India: Byword Publishers; 2005:553-568.
12. Charlson F, Siskind D, Doi SA, et al. ECT efficacy and treatment course: a systematic review and meta-analysis of twice vs thrice weekly schedules. J Affect Disord. 2012;138(1-2):1-8. PubMed doi:10.1016/j.jad.2011.03.039
13. Chanpattana W, Chakrabhand ML, Sackeim HA, et al. Continuation ECT in treatment-resistant schizophrenia: a controlled study. J ECT. 1999;15(3):178-192. PubMed doi:10.1097/00124509-199909000-00002
14. Yoosefi A, Sepehri AS, Kargar M, et al. Comparing effects of ketamine and thiopental administration during electroconvulsive therapy in patients with major depressive disorder: a randomized, double-blind study. J ECT. 2014;30(1):15-21. PubMed doi:10.1097/YCT.0b013e3182a4b4c6
15. Rasmussen KG, Kung S, Lapid MI, et al. A randomized comparison of ketamine versus methohexital anesthesia in electroconvulsive therapy. Psychiatry Res. 2014;215(2):362-365. PubMed doi:10.1016/j.psychres.2013.12.027
16. Sudha S, Andrade C, Mukundan CR, et al. Spectral EEG effects of electroconvulsive shock stimulus parameters: the development of a rationale for the optimization of the ECT stimulus. J ECT. 2003;19(4):197-210. PubMed doi:10.1097/00124509-200312000-00005
17. O’ Donovan S, Kennedy M, Guinan B, et al. A comparison of brief pulse and ultrabrief pulse electroconvulsive stimulation on rodent brain and behaviour. Prog Neuropsychopharmacol Biol Psychiatry. 2012;37(1):147-152. PubMed doi:10.1016/j.pnpbp.2011.12.012
18. Khaleel N, Roopa R, Smitha JS, et al. Electroconvulsive therapy attenuates dendritic arborization in the basolateral amygdala. J ECT. 2013;29(3):156-157. PubMed doi:10.1097/YCT.0b013e318282a6b1
19. Khaleel N, Ravindranath R, Sagar BK, et al. Images in electroconvulsive therapy: pilot impressions suggesting that ECT reduces excitatory synapses in the basolateral amygdala. Indian J Psychiatry. 2013;55(2):204-205. PubMed doi:10.4103/0019-5545.111471
20. Spaans HP, Verwijk E, Comijs HC, et al. Efficacy and cognitive side effects after brief pulse and ultrabrief pulse right unilateral electroconvulsive therapy for major depression: a randomized, double-blind, controlled study. J Clin Psychiatry. 2013;74(11):e1029-e1036. PubMed doi:10.4088/JCP.13m08538
21. Pittenger C, Duman RS. Stress, depression, and neuroplasticity: a convergence of mechanisms. Neuropsychopharmacology. 2008;33(1):88-109. PubMed doi:10.1038/sj.npp.1301574
22. Andrade C, Rao NS. How antidepressant drugs act: a primer on neuroplasticity as the eventual mediator of antidepressant efficacy. Indian J Psychiatry. 2010;52(4):378-386. PubMed doi:10.4103/0019-5545.74318
23. Bouckaert F, Sienaert P, Obbels J, et al. ECT: its brain enabling effects: a review of electroconvulsive therapy-induced structural brain plasticity. J ECT. In Press.
24. Grover S, Mattoo SK, Gupta N. Theories on mechanism of action of electroconvulsive therapy. German J Psychiatry. 2005;8:70-84. http://www.gjpsy.uni-goettingen.de/gjp-article-grover2-ECT.pdf. Updated August 25, 2005. Accessed on May 02, 2014.
25. McCall WV, Andrade C, Sienaert P. Searching for the mechanism(s) of ECT’s therapeutic effect [published online ahead of print April 21, 2014]. J ECT. PubMed
26. Andrade C. Assessment of the therapeutic adequacy of the ECT seizure: current status. NIMHANS J. 1997;15:319-329.
27. Sackeim HA. The anticonvulsant hypothesis of the mechanisms of action of ECT: current status. J ECT. 1999;15(1):5-26. PubMed doi:10.1097/00124509-199903000-00003
28. Mayur P. Ictal electroencephalographic characteristics during electroconvulsive therapy: a review of determination and clinical relevance. J ECT. 2006;22(3):213-217. PubMed doi:10.1097/01.yct.0000235922.14623.39
29. Farzan F, Boutros NN, Blumberger DM, et al. What does the electroencephalogram tell us about the mechanisms of action of ECT in major depressive disorders? J ECT. In Press.
30. Fochtmann LJ. Animal studies of electroconvulsive therapy: foundations for future research. Psychopharmacol Bull. 1994;30(3):321-444. PubMed
31. Mann JJ. Neurobiological correlates of the antidepressant action of electroconvulsive therapy. J ECT. 1998;14(3):172-180. PubMed doi:10.1097/00124509-199809000-00003
32. Haskett RF. Neuroendocrine hypotheses, including pituitary-adrenal axis. J ECT. In Press.
33. Baldinger P, Lotan A, Frey R, et al. Neurotransmitters and ECT. J ECT. In Press.
34. Altar CA, Laeng P, Jurata LW, et al. Electroconvulsive seizures regulate gene expression of distinct neurotrophic signaling pathways. J Neurosci. 2004;24(11):2667-2677. PubMed doi:10.1523/JNEUROSCI.5377-03.2004
35. Chen F, Madsen TM, Wegener G, et al. Repeated electroconvulsive seizures increase the total number of synapses in adult male rat hippocampus. Eur Neuropsychopharmacol. 2009;19(5):329-338. PubMed doi:10.1016/j.euroneuro.2008.12.007
36. Smitha JSM, Roopa R, Khaleel N, et al. Images in ECT: ECS dose-dependently increases dendritic arborization in the CA1 region of the rat hippocampus. J ECT. In Press.
37. Smitha JSM, Roopa R, Sagar BKC, et al. Images in ECT: ECS dose-dependently increases cell proliferation in the subgranular region of the rat hippocampus. J ECT. In Press.
38. Andrade C, Bolwig TG. ECT, hypertensive surge, blood-brain barrier breach, and amnesia: exploring the evidence for a connection. J ECT. In Press.
39. Guloksuz S, Rutten BP, Arts B, et al. The immune system and electroconvulsive therapy for depression [published online ahead of print April 21, 2014]. J ECT.
40. Margoob MA, Ali Z, Andrade C. Efficacy of ECT in chronic, severe, antidepressant- and CBT-refractory PTSD: an open, prospective study. Brain Stimulat. 2010;3(1):28-35. PubMed doi:10.1016/j.brs.2009.04.005

 Each month in his online column, Dr Andrade considers theoretical and practical ideas in clinical psychopharmacology with a view to update the knowledge and skills of medical practitioners who treat patients with psychiatric conditions.
Each month in his online column, Dr Andrade considers theoretical and practical ideas in clinical psychopharmacology with a view to update the knowledge and skills of medical practitioners who treat patients with psychiatric conditions.