ABSTRACT
Objective: To investigate pharmacist-led dementia care rounds (PDRs) and their effect on the use of sleep medications, including the number and content of prescription suggestions during PDRs and use of sleep medications at the time of hospitalization and discharge.
Methods: This was a retrospective observational study of inpatients who received PDR intervention at a hospital in Japan from January 1 to December 31, 2020. The PDR team, consisting of a pharmacist and dementia care nurse, made prescription suggestions through the attending nurse, and the attending physician made the decision to change the prescription. Use of sleep medication was investigated by classifying patients into 2 groups: those for whom prescription suggestions from PDRs were accepted and those for whom they were rejected.
Results: PDRs were conducted 1,164 times with 418 patients, and prescription suggestions were made 330 times (28.4%) for 173 (41.4%) patients. Of these, 234 (70.9%) prescription suggestions were accepted. At the time of discharge, the percentage of patients using benzodiazepine-based sleep medications was 3.1% in the accepted group and 11.9% in the rejected group. The percentage of patients using non–benzodiazepine-based sleep medications was 22.1% in the accepted group and 9.5% in the rejected group. Further, the percentage of patients using non–γ-aminobutyric acid receptor agonist drugs as sleep medications was 9.2% in the accepted group and 2.4% in the rejected group. The results show that the percentage of patients using benzodiazepine-based sleep medications was significantly lower in the accepted group than in the rejected group (P = .022).
Conclusions: PDR intervention contributed to the appropriate use of sleep medications, with nearly 30% of prescription suggestions. PDRs may play an important role in the appropriate use of sleep medications, and active participation of pharmacists in dementia care is necessary.
Prim Care Companion CNS Disord 2022;24(6):22m03258
To cite: Mori K, Yoshida M, Tomita K, et al. Effects of pharmacist-led dementia care round interventions on the use of sleep medications. Prim Care Companion CNS Disord. 2022;24(6):22m03258.
To share: https://doi.org/10.4088/PCC.22m03258
© 2022 Physicians Postgraduate Press, Inc.
aDepartment of Pharmacy, Ogaki Municipal Hospital, Ogaki-shi, Gifu, Japan
bDepartment of Nursing, Ogaki Municipal Hospital, Ogaki-shi, Gifu, Japan
cDepartment of Psychiatric Neurology, Ogaki Municipal Hospital, Ogaki-shi, Gifu, Japan
dLaboratory of Drug Informatics, Gifu Pharmaceutical University, Ogaki-shi, Gifu, Japan
*Corresponding author: Koki Mori, BPharm, Department of Pharmacy, Ogaki Municipal Hospital, 4-86 Minaminokawa-cho, Ogaki-shi, Gifu 503-8502, Japan ([email protected]).
Fifteen percent of people in Japan aged ≥ 65 years have dementia1; therefore, it is necessary to expand medical care, welfare services, and community support for these patients. Patients with dementia often show behavioral and psychological symptoms of dementia (BPSD) due to changes in their living environment associated with hospitalization. Since BPSD is a burden on patients and medical staff,2,3 it is necessary for the medical care team to provide interventions for these patients. The Ogaki Municipal Hospital in Japan established a dementia support team to provide such interventions for patients with BPSD. The dementia support team conducts multidisciplinary rounds (MDRs) for dementia care. This team includes health care experts such as pharmacists, physiotherapists, occupational therapists, speech therapists, and nutritionists among others. In recent years, the importance of pharmacists within the support team conducting MDRs has been reported.4–6
While MDR allows for multifaceted interventions with patients, the multidisciplinary team can only intervene with some of them. With the increasing prevalence of dementia,1 the dementia care teams need to intervene with many patients. Therefore, we created pharmacist-led dementia care rounds (PDRs) with a team consisting of a pharmacist and a dementia care nurse. Older adults are at high risk of adverse drug reactions due to their decreased metabolic capacity7 and polypharmacy.8 Therefore, safe pharmacotherapy is required. We believe that PDR can make a significant contribution toward providing appropriate pharmacotherapy.
Dementia care rounds address a wide range of issues in medical practice. Elderly individuals, in particular, tend to have shorter sleep duration,9 and a change in the environment due to hospitalization often leads to insomnia.10 Therefore, advice is needed when providing care for patients experiencing symptoms such as insomnia and night delirium. Pharmacotherapy for insomnia includes the use of sleep medications. Sleep medications such as benzodiazepines (BZDs) are not recommended for the elderly due to high risk of adverse effects,11 and nonbenzodiazepines (n-BZDs) are a safer option.12 In addition, ramelteon and suvorexant, which do not target the γ-aminobutyric acid (GABA) receptors (also known as non-GABA agonist drugs [n-GADs]), are also safe.13 The purpose of PDR is to promote the appropriate use of sleep medications by avoiding the administration of BZDs and to ensure sleep duration using n-BZD and n-GAD medications.
In this study, we calculated the number and content of prescription suggestions during PDRs and investigated the use of sleep medications at the time of hospitalization and discharge to determine the effect of PDR intervention.
METHODS
We conducted a retrospective observational analysis of PDR interventions and the effect on prescribing of medications to inpatients at Ogaki Municipal Hospital between January 1 and December 31, 2020. We investigated the PDRs in terms of the number, content, and acceptance rate of prescription suggestions using data available in the electronic medical records. This study was approved by the Institutional Review Board of Ogaki Municipal Hospital.
Sample
Patients who received PDR intervention were included in the study, and those who died during hospitalization were excluded. The data on PDR interventions and prescription details were collected from the electronic medical records.
PDR Procedure
Nurses request dementia care round interventions for inpatients with dementia and delirium, and the dementia care nurse receives the request. The dementia care nurse assigns patients who require pharmacist intervention to the PDR. The pharmacist and dementia care nurse then visit the ward. The pharmacist collects the relevant information from the electronic medical records and the attending nurses. The dementia care nurse provides advice to the attending nurses to improve environmental arrangements and patient supports in the electronic medical record and verbally. The pharmacist reviews the drug therapy and, if necessary, makes prescription suggestions to the attending physician through the attending nurses and the electronic medical record. The pharmacist and the dementia care nurse then visit the patient and check their condition, including level of wakefulness and speech. This activity is conducted once a week during the daytime between 1:00 pm and 4:00 pm.
Data
Patient backgrounds and other details regarding their hospitalization were collected retrospectively from electronic medical records retrieved from the department of hospitalization. Patient background data included age, sex, height, weight, body mass index, sequence of events during hospitalization to discharge (days of hospitalization, days from intervention to discharge, number of inpatients coming from their home, and number of patients discharged to home), number of inpatients by department (medicine or surgery), and the type of dementia and long-term care requirement certification.
Data were retrieved on the use of sleep medications (BZD, n-BZD, n-GAD) at the time of hospitalization and discharge, change of BZD use (discontinuation of BZD, addition to BZD), number of medications (at hospitalization, at discharge, and change in number of drugs), sleep duration (3 days after hospitalization, 3 days before discharge, and average change), patients with physical restraint (started before intervention, after intervention, and at the time of discharge), and occurrence of falls (before and after intervention). Drug types were classified based on the Anatomical Therapeutic Chemical Classification System. Benzodiazepine derivatives were classified as BZD, BZD-related drugs as n-BZD, and ramelteon and suvorexant as n-GAD. We defined antihistamines, narcotics, and corticosteroids as delirium risk drugs based on previous reports.14
We tabulated the number and content of PDRs from January to December 2020. The main content included the number of rounds and the number of prescription suggestions. Prescription suggestions were categorized by drug type suggested (antidementia drugs, antipsychotics, antidepressants, sleep medications, delirium risk drugs, others), content of suggestions (addition, dose change, discontinuation, usage change, others), and the number of accepted prescription suggestions. We classified patients whose attending physician accepted the prescription suggestions at least once as the accepted group and those who rejected all suggestions as the rejected group.
Outcomes
The primary endpoint of this study was the proportion of sleep medications (BZD, n-BZD, n-GAD) used at the time of hospitalization and discharge. The secondary endpoints of this study were changes in the number of drugs, changes in sleep duration, presence of physical restraints, and presence of falls. The change in the number of drugs was defined as the difference between the number of drugs at the time of hospitalization and discharge. The change in sleep duration was defined as the difference between the average sleep time during the 3 days immediately after hospitalization and the average sleep time during the 3 days immediately before discharge.
Data Analysis
The differences in continuous data were compared using the Mann-Whitney U test, and differences in categorical data were compared using Fisher exact test or χ2 test. In all statistical analyses, the level of significance was set at .05.
RESULTS
Outline of PDR Procedure
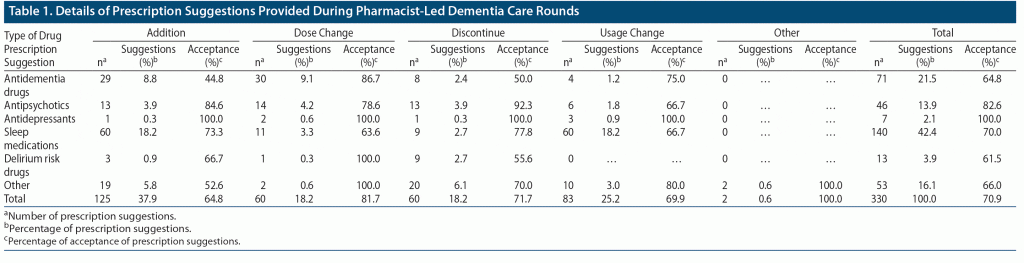
PDRs were performed 1,164 times for 418 patients in a year. Prescription suggestions were made in 330 (28.4%) PDRs performed for 173 (41.4%) patients. Of the 330 prescription suggestions made, 234 (70.9%) were accepted. Table 1 shows the breakdown of the prescription suggestions provided. The most common prescription suggestions, given to 18.2% of the patients, respectively, were the addition of sleep medications and usage change. These prescriptions were followed by dosage change (9.1%) and addition (8.8%) of antidementia drugs.
Effect of PDR
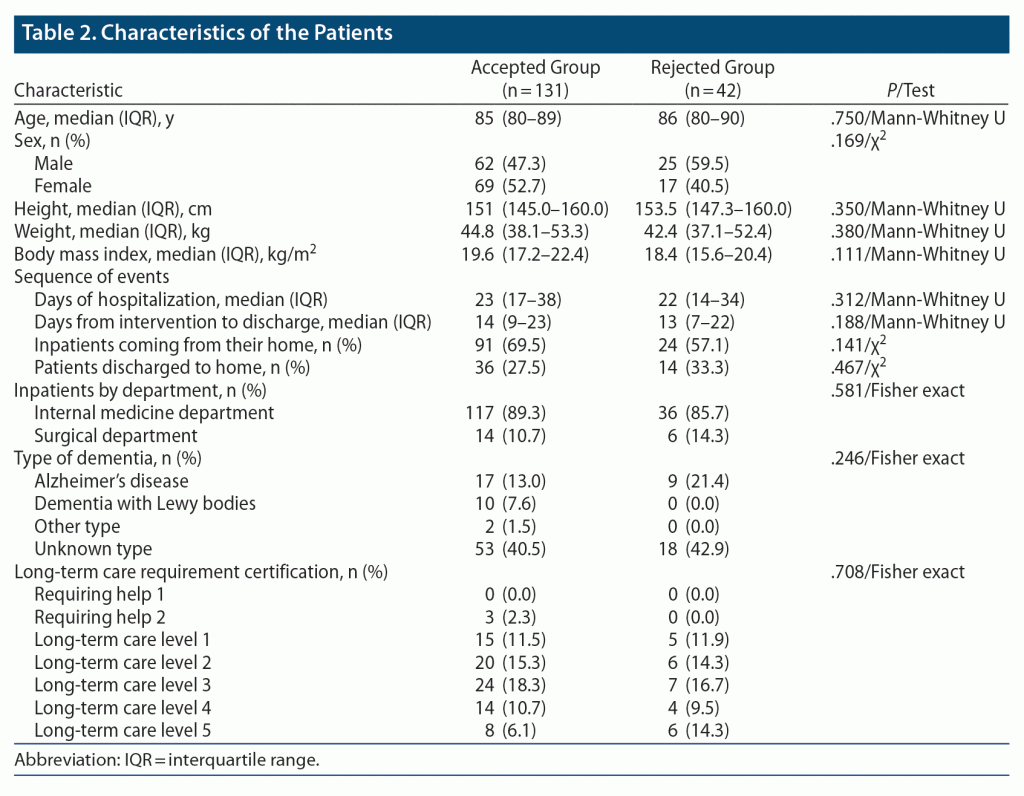
Table 2 presents the background information for the patients. There were 173 patients: 131 in the accepted group and 42 in the rejected group. In terms of patient background, there was no significant difference between the accepted and rejected groups.
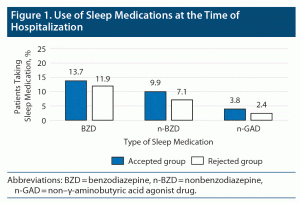
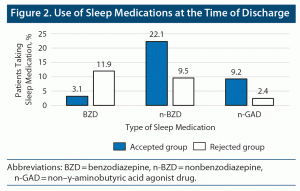
With respect to the primary endpoint of the study, there was no significant difference between the rates of participants from the accepted and rejected groups regarding the use of sleep medications during hospitalization (P = .949) (Figure 1). However, there were significant differences in the rates of patients using sleep medications at discharge. The rate of patients in the accepted group using BZDs decreased, and those using n-BZDs and n-GADs increased (P = .022) (Figure 2).
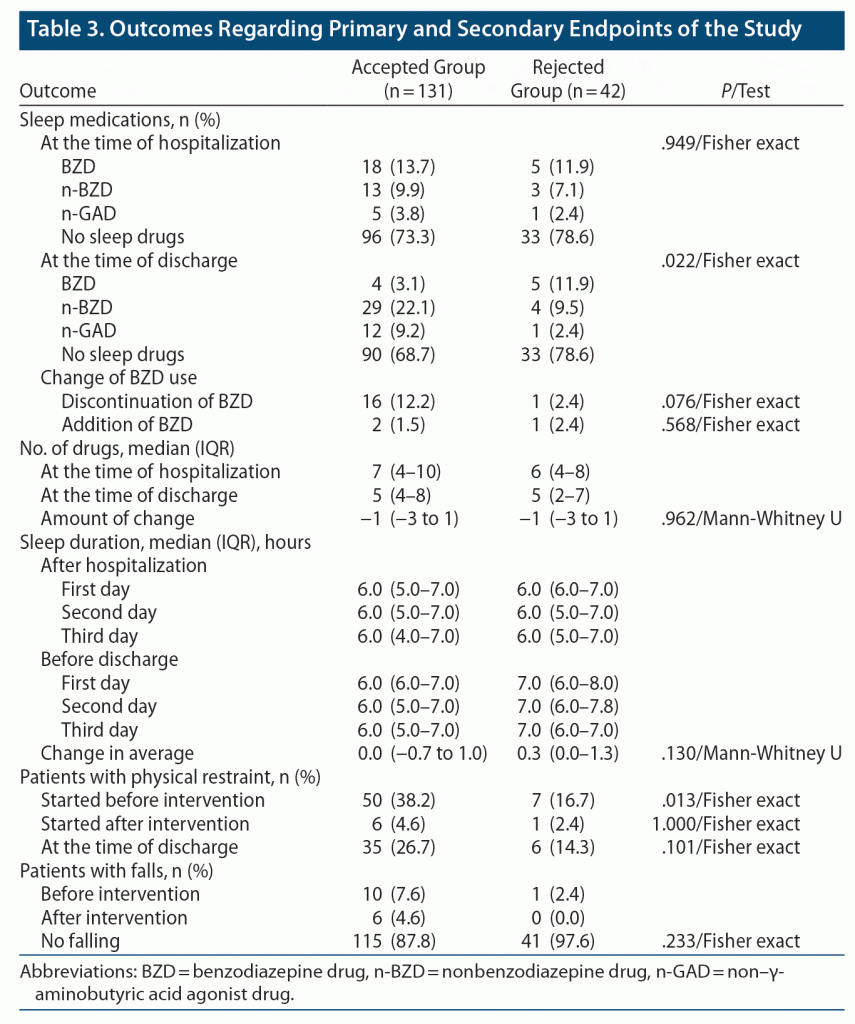
With respect to the secondary endpoints of the study, the use of physical restraint started before intervention was significantly higher in the accepted group compared with the rejected group (P = .013). There were no significant differences between the accepted and rejected groups for the other items (Table 3).
DISCUSSION
In this study, PDR contributed to the administration of many interventions, mainly regarding sleep medications, with nearly 70% acceptance. The results also showed that PDR may contribute to the reduction of BZDs at the time of hospital discharge. These results suggest that PDR is a useful technique for administering interventions.
PDR is a round of visits conducted by professionals of only 2 occupations: pharmacists and nurses. Rounds with fewer professionals involved in fewer occupations can lead to an increase in the number of interventions with less effort. More than 1,000 PDRs were performed within the study period, with 40% of patients receiving interventions and 30% receiving prescription suggestions. Further, the acceptance rate was approximately 70%. PDR interventions, when categorized by drug type, mostly comprised interventions involving sleep medications and antidementia drugs. In terms of content of suggestions provided by PDR, most interventions involved addition of drugs and changes in drug dose. The elderly are prone to insomnia,10 and appropriate use of sleep medications is also necessary.11,15 Therefore, PDR was able to provide interventions regarding sleep medication for many patients. Interventions administered using antidementia drugs mainly involved addition of these drugs for patients with untreated dementia and dose change, which is similar to the outcomes of a previous MDR report.5
Regarding the effect of PDR, the use of BZDs at discharge was significantly lower in the accepted group. Since the use of sleep medications during hospitalization was similar between the 2 groups, it is assumed that the reduction in the rate of use of BZDs was due to the PDR intervention. BZDs are not recommended for the elderly11 due to the high risk of cognitive decline16 and falls.17 Thus, use of n-BZDs and n-GADs has increased. While n-BZDs are safer than BZDs,12 there is a risk of falls due to their stimulating effect on the GABA receptor, a mechanism that is similar to that of BZDs.18 n-GADs have a low risk of falls13 and prevent delirium.19,20 Thus, use of n-GADs is safe for the elderly. However, patient satisfaction is low.15 Continued insomnia is associated with increased delirium21 and mortality.22 Therefore, it is important to ensure proper sleep duration. In this study, the change in average sleep duration was similar for patients in the 2 groups, even though there was a difference in sleep medications between them. The PDR interventions thus avoided BZD use but still ensured sleep duration with other drugs. Therefore, it is unlikely that the increased use of n-BZDs and n-GADs will unilaterally disadvantage the elderly.
Use of physical restraint may be clinically unavoidable in the provision of safe medical care. However, physical restraint is associated with a risk of delirium,23 gait disturbance, falls, and hearing impairment.24 Therefore, it is important to avoid physical restraints in dementia care. In this study, we investigated the rate of physical restraint as a secondary outcome. The rate of patients with physical restraint started before intervention was higher in the accepted group compared to the rejected group, with no significant difference after intervention and at discharge. The difference in physical restraint before intervention may be due to more restless patients in the PDR group, which thus led to the acceptance of the prescription suggestion. It is unlikely that PDR affected physical restraint because the patients with physical restraint were of a similar ratio after intervention and at discharge. There were no significant differences in the other endpoints between the 2 groups.
One limitation of this study was that it did not assess quality of life or other cognitive functions of the patients. Dementia care is based on person-centered care and aims to improve quality of life and cognitive function.25 However, because of the retrospective nature of the study, we were unable to investigate these variables. Another limitation is the lack of investigation into the use of sleep medications after discharge. Cognitive decline is a risk factor associated with long-term use of BZDs,16 and long-term use of this class of drugs should be avoided. Because most of the patients were transferred to another hospital or health care facility, we were unable to investigate their use of sleep medications after discharge. Another limitation is that we were unable to show the effects of dementia care nurse intervention. Although this study focused only on pharmacotherapy, the advice for patient care is also important. But because the patients were classified according to their acceptance of prescription suggestions, we could not perform the appropriate evaluation of the dementia care nurse intervention.
In this study, PDRs were performed with many patients, and their contribution to appropriate pharmacotherapy was confirmed. In other words, pharmacists should be involved in both MDR and PDR activities. PDR may play an important role in the appropriate use of sleep medications, and active participation of pharmacists in dementia care is necessary.
Submitted: February 4, 2022; accepted May 11, 2022.
Published online: November 29, 2022.
Relevant financial relationships: None.
Funding/support: None.
Clinical Points
- Pharmacist-led dementia care rounds (PDRs) are an efficient way to correct sleep medication use.
- PDR allows for many patient rounds and pharmacologic interventions.
- PDR can contribute to benzodiazepine discontinuation, which is especially important in the elderly.
References (25)

- Ninomiya T, Nakaji S, Maeda T, et al; JPSFC-AD Study Group. Study design and baseline characteristics of a population-based prospective cohort study of dementia in Japan: the Japan Prospective Studies Collaboration for Aging and Dementia (JPSC-AD). Environ Health Prev Med. 2020;25(1):64–75. PubMed CrossRef
- Matsumoto N, Ikeda M, Fukuhara R, et al. Caregiver burden associated with behavioral and psychological symptoms of dementia in elderly people in the local community. Dement Geriatr Cogn Disord. 2007;23(4):219–224. PubMed CrossRef
- Cerejeira J, Lagarto L, Mukaetova-Ladinska EB. Behavioral and psychological symptoms of dementia. Front Neurol. 2012;3:73. PubMed
- Tan ZS, Damron-Rodriguez J, Cadogan M, et al. Team-based interprofessional competency training for dementia screening and management. J Am Geriatr Soc. 2017;65(1):207–211. PubMed CrossRef
- Mori K, Yoshida M, Tomita K, et al. Intervention of dementia support team pharmacists and its effects for the proper use of rivastigmine. Pharmazie. 2021;76(7):334–337. PubMed
- Gustafsson M, Sjölander M, Pfister B, et al. Pharmacist participation in hospital ward teams and hospital readmission rates among people with dementia: a randomized controlled trial. Eur J Clin Pharmacol. 2017;73(7):827–835. PubMed CrossRef
- O’Malley K, Crooks J, Duke E, et al. Effect of age and sex on human drug metabolism. BMJ. 1971;3(5775):607–609. PubMed CrossRef
- Kojima T, Akishita M, Kameyama Y, et al. High risk of adverse drug reactions in elderly patients taking six or more drugs: analysis of inpatient database. Geriatr Gerontol Int. 2012;12(4):761–762. PubMed CrossRef
- Ohayon MM, Carskadon MA, Guilleminault C, et al. Meta-analysis of quantitative sleep parameters from childhood to old age in healthy individuals: developing normative sleep values across the human lifespan. Sleep. 2004;27(7):1255–1273. PubMed CrossRef
- Miller MA, Renn BN, Chu F, et al. Sleepless in the hospital: a systematic review of non-pharmacological sleep interventions. Gen Hosp Psychiatry. 2019;59:58–66. PubMed CrossRef
- Fick DM, Semla TP, Steinman M, et al; By the 2019 American Geriatrics Society Beers Criteria Update Expert Panel. American Geriatrics Society 2019 Updated AGS Beers Criteria for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2019;67(4):674–694. PubMed CrossRef
- Schroeck JL, Ford J, Conway EL, et al. Review of safety and efficacy of sleep medicines in older adults. Clin Ther. 2016;38(11):2340–2372. PubMed CrossRef
- Torii H, Ando M, Tomita H, et al. Association of hypnotic drug use with fall incidents in hospitalized elderly patients: a case-crossover study. Biol Pharm Bull. 2020;43(6):925–931. PubMed CrossRef
- Tuma R, DeAngelis LM. Altered mental status in patients with cancer. Arch Neurol. 2000;57(12):1727–1731. PubMed CrossRef
- Borchert JS, Wang B, Ramzanali M, et al. Adverse events due to insomnia drugs reported in a regulatory database and online patient reviews: Comparative study. J Med Internet Res. 2019;21(11):e13371. PubMed CrossRef
- He Q, Chen X, Wu T, et al. Risk of dementia in long-term benzodiazepine users: evidence from a meta-analysis of observational studies. J Clin Neurol. 2019;15(1):9–19. PubMed CrossRef
- Seppala LJ, Wermelink AMAT, de Vries M, et al; EUGMS task and Finish group on fall-risk-increasing drugs. Fall-risk-increasing drugs: a systematic review and meta-analysis: II. Psychotropics. J Am Med Dir Assoc. 2018;19(4):371.e11–371.e17. PubMed CrossRef
- Yu NW, Chen PJ, Tsai HJ, et al. Association of benzodiazepine and Z-drug use with the risk of hospitalization for fall-related injuries among older people: a nationwide nested case-control study in Taiwan. BMC Geriatr. 2017;17(1):140. PubMed CrossRef
- Wu YC, Tseng PT, Tu YK, et al. Association of delirium response and safety of pharmacological interventions for the management and prevention of delirium: a network meta-analysis. JAMA Psychiatry. 2019;76(5):526–535. PubMed CrossRef
- Hatta K, Kishi Y, Wada K, et al. Real-world effectiveness of ramelteon and suvorexant for delirium prevention in 948 patients with delirium risk factors. J Clin Psychiatry. 2019;81(1):19m12865. PubMed
- Trompeo AC, Vidi Y, Locane MD, et al. Sleep disturbances in the critically ill patients: role of delirium and sedative agents. Minerva Anestesiol. 2011;77(6):604–612. PubMed
- Ikehara S, Iso H, Date C, et al; JACC Study Group. Association of sleep duration with mortality from cardiovascular disease and other causes for Japanese men and women: the JACC study. Sleep. 2009;32(3):295–301. PubMed CrossRef
- Pan Y, Jiang Z, Yuan C, et al. Influence of physical restraint on delirium of adult patients in ICU: a nested case-control study. J Clin Nurs. 2018;27(9-10):1950–1957. PubMed CrossRef
- Fernández Ibáñez JM, Morales Ballesteros MDC, Montiel Moreno M, et al. Physical restraint use in relation to falls risk in a nursing home. Rev Esp Geriatr Gerontol. 2020;55(1):3–10. PubMed
- Clissett P, Porock D, Harwood RH, et al. The challenges of achieving person-centred care in acute hospitals: a qualitative study of people with dementia and their families. Int J Nurs Stud. 2013;50(11):1495–1503. PubMed CrossRef
Please sign in or purchase this PDF for $40.
Save
Cite