LESSONS LEARNED AT THE INTERFACE OF MEDICINE AND PSYCHIATRY
The Psychiatric Consultation Service at Massachusetts General Hospital sees medical and surgical inpatients with comorbid psychiatric symptoms and conditions. During their twice-weekly rounds, Dr Stern and other members of the Consultation Service discuss diagnosis and management of hospitalized patients with complex medical or surgical problems who also demonstrate psychiatric symptoms or conditions. These discussions have given rise to rounds reports that will prove useful for clinicians practicing at the interface of medicine and psychiatry.
Prim Care Companion CNS Disord 2022;24(3):21f02958
To cite: Van Alphen MU, Paudel S, Vyas CM, et al. Facilitating smoking cessation in primary care settings. Prim Care Companion CNS Disord. 2022;24(3):21f02958.
To share: https://doi.org/10.4088/PCC.21f02958
© Copyright 2022 Physicians Postgraduate Press, Inc.
aDepartment of Psychiatry, Massachusetts General Hospital, Boston, Massachusetts
bHarvard Medical School, Boston, Massachusetts
cNorth Suffolk Mental Health Association, Boston, Massachusetts
dDepartment of Internal Medicine, The BronxCare Health System, Bronx, New York
‡Drs Van Alphen, Paudel, Schnitzer, Vyas, and Patel and Ms Mulligan contributed equally to this work.
*Corresponding author: Manjola U. Van Alphen, MD, PhD, MBA, Department of Psychiatry, Massachusetts General Hospital, Harvard Medical School, 25 Staniford St, Boston, Massachusetts ([email protected]).
Have you been uncertain about whether you should ask your patients about their smoking habits? Have you felt confident in your ability to help your patients stop smoking? Have you puzzled over whether you should provide them with information, a prescription to reduce nicotine cravings, or guidance regarding behavioral health strategies? Have you been uncertain about safe prescribing practices for patients with co-occurring mental illness? If you have, the following case vignette and discussion should enhance your understanding of the importance of tobacco cessation, best practices (involving pharmacologic agents and counseling techniques), and strategies to address myths related to smoking cessation.
CASE VIGNETTE
Mr A, a 30-year-old college graduate disabled by schizophrenia (manifest by auditory hallucinations), wanted to discuss options for smoking cessation with his physician. He started smoking cigarettes at the age of 15 years and had been smoking 1.5 packs/day for the past 5 years. Although he noted that smoking relaxed him, he had tried to quit several times without success. He used a nicotine patch and gum intermittently but felt that they were of little benefit. He regretted spending so much of his money on cigarettes every month. He did not drink alcohol, smoke marijuana, or use other recreational substances. He reported no delusions, depressed mood, euphoria, or recent changes in his energy level, appetite, sleep, or motivation. He felt safe and supported and had no thoughts of self-harm.
On examination, Mr A was overweight, appropriately dressed, well groomed, calm, cooperative, and fully oriented with a euthymic mood and a blunted affect. He appeared to respond to internal stimuli (by looking around and smiling episodically). His thought process was linear, and he had no thoughts of violence. His decisions around treatment were appropriate. His recent laboratory tests revealed mildly elevated total and low-density lipoprotein cholesterol levels, while the complete blood count, comprehensive metabolic panel, and hemoglobin A1c results were within the normal range.
Mr A’s physician appreciated his efforts to quit smoking, acknowledged the perceived benefits, and emphasized the importance of quitting. He discussed behavioral approaches and pharmacologic cessation aids, including nicotine replacement therapy (NRT), bupropion (Wellbutrin), and varenicline (Chantix). Mr A was interested in taking a medication; however, he was concerned about the “scary side effects” of varenicline for those with mental illness. His physician informed him about the safety and efficacy of varenicline and the evidence that varenicline is safe for people with mental illness and helps to ameliorate withdrawal symptoms. However, Mr A was still unwilling to start varenicline at this point; instead, he asked if he could try bupropion first. He was started on sustained-release bupropion (Wellbutrin SR) 150 mg once/day with a plan to increase the dose to 150 mg twice/day after 1 week.
During a follow-up appointment 2 months later, Mr A reported that he was still smoking about 1 pack/day and had received little benefit from use of bupropion. He then agreed to try varenicline. It was decided that he would take varenicline for 1 month before attempting to reduce or stop smoking cigarettes. His physician wrote a prescription for a starter pack, beginning with 0.5 mg once/day, working up to a dose of 1 mg twice/day. His physician explained that stomach upset and difficulty with sleep were the most common side effects, but that they typically abated over time, and discussed strategies to mitigate these side effects (eg, taking it with food, taking the nighttime dose earlier, undergoing a slower uptitration if needed). After 2 months on varenicline, Mr A had reduced his cigarette smoking to a half pack/day, and he was hopeful that he would be able to quit. At their next appointment, his physician planned to suggest the addition of short- and long-acting NRT to assist in Mr A’s cessation attempt.
DISCUSSION
Why Is It Important for Your Patients (and their family members) to Cut Back or Stop Cigarette Smoking?
Who doesn’t want to save money and live better and longer? Simply put, cutting back or stopping cigarette smoking can improve the overall quality of health and life for patients and their family members, while increasing overall life expectancy by a decade and putting extra dollars in their pockets.1,2
The perils of smoking tobacco and thus the importance of quitting have been the focus of investigations since the mid-20th century,3 as tobacco smoking continues to be the single leading cause of preventable disease, disability, and premature death in the United States and worldwide.1,4 Tobacco smoking contributes to at least 12 types of cancer including lung cancer and other forms of cancer (cancers of the larynx, oral cavity and pharynx, esophagus, pancreas, bladder, stomach, liver, colon and rectum, kidney, and cervix and acute myeloid leukemia) and several chronic diseases (including cardiovascular, cerebrovascular, and pulmonary diseases and metabolic, neurologic, psychiatric, substance use, fertility, and perinatal disorders). Second-hand smoking, also known as passive smoking, is especially hazardous for pregnant women and children, and it affects 40% of all children, 35% of women, and 33% of men in the general population.1,3,4 Second-hand smoking increases the risk of lung cancer and cardiovascular disease in adults and contributes to ear and lung disease in children and to sudden infant death syndrome.
Tobacco smoking is associated with up to a 70% increase in age-specific death rates, leading to an estimated loss of a decade of life.3 Patients with serious mental illness (SMI) such as schizophrenia have a 25- to 30-year mortality gap, with tobacco smoke as a primary contributor.5
Although the prevalence of current cigarette smoking among US adults in 2018 was at a historical low of 13.8% (marking a decline of more than a third from the 1965 reported rate of 20.9%), the relative risk of all-cause mortality from smoking has increased in both men (from 1.76 to 2.33) and women (from 1.35 to 2.08) during the same time interval.1,3 This accelerated risk of death in smokers over these 50 years has been primarily attributed to heavier lifetime smoking, especially in generations who began smoking in their teens and continued to smoke into middle and older ages.3 While the relative risk of dying from smoking has increased over time, the benefits of quitting persist, with a decrease in excess mortality ranging from 40% when quitting smoking by 60 years of age to 90% when quitting smoking by age 40 years.1 Furthermore, quality of health and overall quality of life increase significantly with smoking cessation.2
Economic costs associated with smoking (for patients, their families, and society at large) are substantial.1,6 On average, smoking 1 pack of cigarettes a day costs $6.28 ($188/month and $2,292/year). Thus, smoking for 10 or 20 years costs a patient and their family an average of $34,318 and $91,671, respectively, in future dollars (adjusted for assumed inflation).6 However, the total economic impact of smoking in the United States has been estimated to exceed $200 billion in current dollars when considering direct costs (ie, inpatient care, outpatient visits, drugs, and diagnostic tests as well as transportation costs), indirect costs (ie, productivity loss, the cost of premature retirement, and morbidity costs), and intangible costs (difficult-to-monetize consequences as pain and suffering and emotional well-being) associated with smoking.1 Economic studies of smoking cessation interventions have consistently demonstrated cost effectiveness and decreased overall costs to patients, families, and society.1
When meeting with patients, providers should provide the above information in layperson’s terms while using motivational interviewing and engagement techniques. This strategy may increase the chances of choosing to stop smoking, although more evidence is needed to support this claim.7
What Signs and Symptoms Can Arise With Cutting Back on or Stopping Use of Nicotine Products?
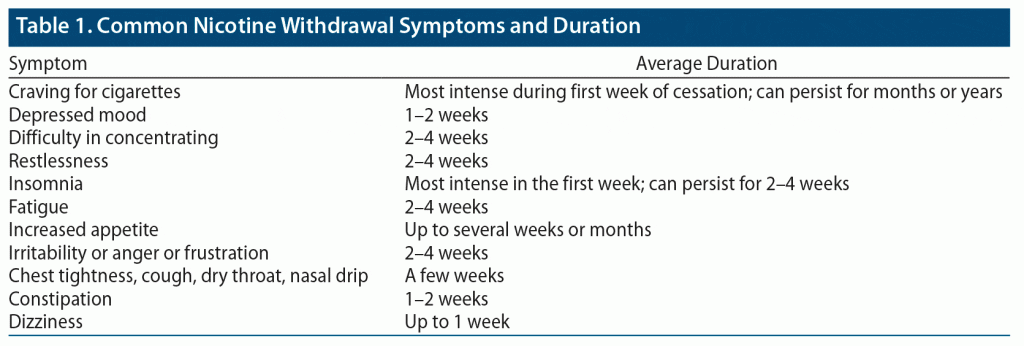
Nicotine withdrawal is manifest by a constellation of physical and psychological symptoms that typically develop within several weeks of stopping or decreasing cigarette smoking. According to the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5), there are 7 primary symptoms associated with nicotine withdrawal: irritability/anger/frustration, anxiety, depressed mood, inability to concentrate, increased appetite, insomnia, and restlessness.8 Moreover, several clinical studies have reported additional physical signs and symptoms (eg, constipation, cough, dizziness, mouth ulcers).9 Of note, depressed mood more frequently occurs in smokers with a history of major depression10–12; the incidence of a new major depressive episode is higher among patients with a history of recurrent depression versus those without major depression.11 Table 1 lists the most common symptoms of nicotine withdrawal and their duration. Symptoms of nicotine withdrawal usually begin within 2 days, peak during the first week, and last 3 to 4 weeks after nicotine use has decreased or stopped. However, some patients experience cravings for cigarettes for an extended duration. Studies13–15 have found that long-term nicotine use interferes with brain functions and neural processes (eg, extended amygdala, dysregulated reward process); these alterations may correlate with elevated nicotine cravings in the abstinence period.16,17
Notably, evidence suggests that cigarette smokers with mental illness experience more severe symptoms of nicotine withdrawal during the first week of a quit attempt than other would-be cigarette quitters.10,18,19 The symptom burden of nicotine withdrawal may be greatest among those with psychotic disorders.20 The severity of withdrawal symptoms is largely determined by the method of nicotine consumption, the frequency of nicotine use, and one’s living conditions (eg, a smoke-free environment). Since more severe symptoms of nicotine withdrawal are associated with smoking relapse, primary care physicians (PCPs) should help their patients navigate nicotine withdrawal symptoms and offer treatment for nicotine withdrawal.20,21 Pharmacogenetic studies have identified molecular mechanisms that govern the emergence and intensity of nicotine withdrawal symptoms.15 Approaches using personalized medicine can be employed to prevent severe withdrawal among those with SMI. Of note, nicotine withdrawal causes symptoms that are similar to common side effects of US Food and Drug Administration (FDA)–approved medications for smoking cessation (eg, bupropion, varenicline) including sleep disturbances. Adding adjunctive approaches (eg, physical activity, mindful-based approaches) can be helpful among smokers with higher levels of sleep deficits.22,23
Which Behavioral Strategies Can Facilitate Smoking Cessation?
While behavioral health interventions and use of medications can facilitate smoking cessation, pharmacotherapy and counseling are most successful when used in tandem.24,25 This is especially true for patients with SMI, as rates of smoking cessation with behavioral treatments alone are only ~4%.24,26
Given the limited time at each visit and the infrequent visits to PCPs, knowledge of available behavioral therapies and the practitioners to whom patients can be referred is crucial, as is the nature of advice/interventions that can be provided. Even brief interventions (less than 3 minutes) can enhance motivation and increase the likelihood of current or future quit attempts.24 Use of the “5As” framework can ensure that every patient is screened for tobacco use routinely; this allows for documentation of tobacco use to be recorded, and current smokers can be given advice on how to quit.24,27 This strategy includes (1) asking all patients about their smoking status and documenting the form, frequency, and severity of use; (2) advising all tobacco users to quit, incorporating clear and personalized information; (3) assessing the willingness to make a quit attempt; (4) assisting in their effort to quit by creating a plan to quit or to reduce cigarette smoking and recommending appropriate pharmacotherapy; and (5) arranging for follow-up, which includes referral to smoking cessation services.25,27 The 5As approach has been shown to have a modest effect in patients with SMI, wherein routine follow-up has aided patients to move through the stages of change (precontemplation, contemplation, preparation, action, and maintenance).28 If patients are unwilling to quit or reduce their smoking, brief motivational interviewing strategies can be employed to explore their beliefs about tobacco use, support self-efficacy, and resolve their ambivalence about behavioral change.27
After utilizing the 5As, referrals can be made for various modalities (including behavioral counseling, involvement of smoking cessation groups, telephone counseling, and use of self-help materials or apps) based on the individual’s needs. Individual behavioral counseling with a tobacco specialist often utilizes cognitive-behavioral processing and mindfulness-based techniques.25 Group behavioral therapy also provides behavioral counseling and peer support.27,29 Telephone counseling often involves Quitlines or Nic A (Nicotine Anonymous) that can be accessed 24/7, requires limited resources, and can be used by underserved populations.25,27 Patients can also be provided with the phone number 1–800-QUIT-NOW (1-800-784-8669) to access free coaching, educational materials, and nicotine patches from services across the nation. SmokefreeTXT is an alternative program that offers free text messaging for 24/7 support. Patients can text QUIT to 47848 to access the service. Self-help materials include manuals, structured programs, and smartphone apps (including QuitSTART by smokefree.gov).27
What Types of Pharmacologic Agents Can Facilitate Smoking Cessation?
PCPs can help their patients recognize the symptoms of nicotine withdrawal and offer treatment with FDA-approved pharmacologic agents to facilitate smoking cessation. An understanding of the neuropathology of nicotine addiction has already opened new frontiers, including effective NRT and use of oral non-nicotine medications (eg, bupropion and varenicline).
Nicotine Replacement Therapy
NRT was the first agent shown to be effective for facilitating smoking cessation. NRT aims to reduce the motivation to smoke cigarettes and to mitigate symptoms of nicotine withdrawal through delivery of low-doses of nicotine, thus easing the transition from cigarette smoking to abstinence.30 NRTs increase the quit rate by 50%–60%; the relative efficacy is largely independent of the additional behavioral support/counseling that is provided to patients.30 Several over-the-counter NRTs are available, including a transdermal patch, gum, and lozenges and 2 prescription-only (microtabs and an inhaler or nasal spray) formulations that are equally effective for smoking cessation.30–32
PCPs should counsel their patients on the appropriate use of over-the-counter products, as many misconceptions about NRT exist. The transdermal nicotine patch delivers nicotine slowly over 24 hours and should be replaced daily. The patch should be placed on a clean, hairless patch of skin between the waist and neck. The patient should also be advised to rotate placement of the patch to minimize risk of skin irritation. The nicotine patch comes in 3 strengths (7 mg, 14 mg, 21 mg) with a dose determined based on a patient’s current tobacco use. For individuals smoking > 40 cigarettes/day, the maximum patch dose of two 21-mg patches (42 mg/day total) should be used. Individuals smoking 31–40 cigarettes/day should be advised to start at 35 mg/day (one 21-mg patch plus one 14-mg patch), those smoking 10–30 cigarettes/day should start with one 21-mg patch, and those smoking < 10 cigarettes/day should start with one 14-mg patch. Once a patient has started using the patch, a reasonable goal is to decrease the dose over a period of 8–12 weeks, though this can be expanded depending on the patient’s current nicotine burden.
Patients with SMI tend to smoke more regularly, display patterns of heavy smoking, extract more nicotine and carbon monoxide from each cigarette, and develop severe nicotine dependence.33–35 Therefore, such patients often require more than 1 patch to achieve significant relief from nicotine withdrawal and may benefit from combining a faster-onset and shorter-duration form of nicotine (such as gum, lozenge, inhaler, or nasal spray).
The EAGLES (Evaluating Adverse Events in a Global Smoking Cessation Study) trial evaluated the relative efficacy and neuropsychiatric safety of smoking cessation medications compared with placebo in smokers with and without SMI.24,36 The transdermal nicotine patch was more effective than placebo, whereas varenicline demonstrated superior efficacy compared to single NRT, bupropion, and placebo.24,36 A combination of a nicotine patch plus a nicotine lozenge, wherein the transdermal patch was applied daily for continuous nicotine delivery and the lozenge was used as needed, was more effective at relieving withdrawal symptoms and cravings than a single formulation of NRT.37,38 Moreover, the use of nicotine patches for up to 24 weeks was found to be safe and effective in primary care practices.39 Commonly reported side effects with any form of NRTs include chest pain, palpitations, gastrointestinal symptoms, and insomnia.40 Skin irritation is a side effect specific to the use of nicotine patches. In addition, mouth or throat soreness, mouth ulcers, hiccoughs, and coughing are side effects specific to orally administered NRTs. Newer products (such as electronic nicotine delivery systems, eg, e-cigarettes and vape pens) are gaining in popularity among smokers. A clinical study41 found a higher abstinence rate among the e-cigarette group versus NRT.
Bupropion
Sustained-release bupropion (under the brand name Wellbutrin SR) is the first FDA-approved non-nicotine medication to facilitate smoking cessation in adults. The recommended starting dose of bupropion SR for smoking cessation is 150 mg once/day (used for 3 days) and then increased to 150 mg twice/day (for 7 to 12 weeks). Importantly, bupropion appears to be relatively ineffective for long-term abstinence in patients with SMI. For example, a recent meta-analysis42 of 28 randomized clinical trials concluded that bupropion increased the likelihood (by 3.5-fold) of smoking cessation at 3 months, especially at a dose of 300 mg/day, but no significant effect was observed at 6 months. Furthermore, the addition of a 7-week bupropion trial to NRT and counseling did not increase the rates of abstinence.43
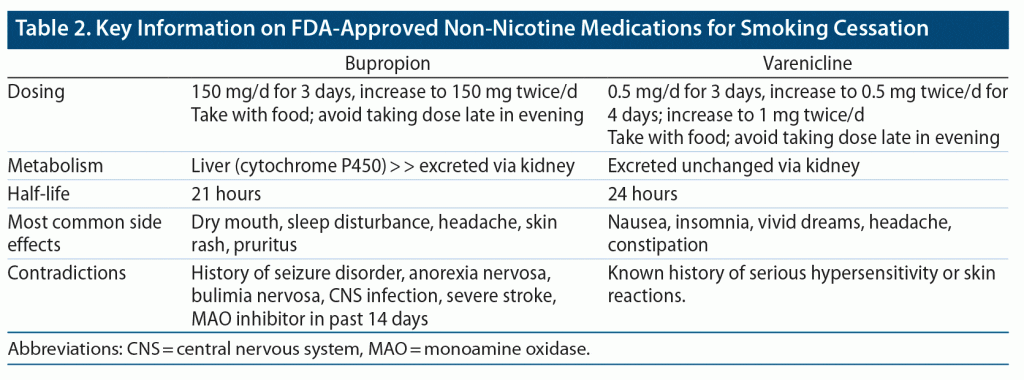
Patients should be prepared for possible side effects before they begin bupropion for smoking cessation. Key information on the use of bupropion in smoking cessation is described in Table 2. The most common side effects of bupropion include dry mouth, insomnia, skin rash, pruritus, and hypersensitivity reactions. To avoid insomnia, patients should not take bupropion after 5 pm. Taking bupropion 1 to 2 weeks before reducing or quitting cigarettes may allow for the establishment of stable therapeutic levels of bupropion; this approach may facilitate the quitting process. Monitoring for seizure, hypertension (only if used with NRT), and worsening and emergence of suicidal thoughts/behaviors is recommended. Serious neuropsychiatric adverse events have been observed in patients with SMI when treated with bupropion for smoking cessation. Substantial concerns have been raised that such neuropsychiatric side effects are related to bupropion. However, a large clinical study16 (EAGLES trial) confirmed that bupropion does not increase the risk of neuropsychiatric adverse events as compared to other agents or placebo.
Varenicline
Varenicline (Chantix) appears to be the most effective pharmacologic agent to facilitate smoking cessation for individuals with and without mental illness. For those with schizophrenia, its use increases the likelihood of quitting by nearly 5-fold.24,36,44 Importantly, varenicline does not increase the risk of adverse neuropsychiatric events as compared to other agents or placebo (per the EAGLES trial),24,36 and a black box warning for it has been removed. Table 2 describes key information on varenicline use for smoking cessation. A starter-pack prescription may be ordered, with dosing starting at 0.5 mg/day and increasing to 1 mg twice/day. In the general population, a typical varenicline trial lasts 12 weeks; however, maintenance doses of varenicline for a year or longer have markedly reduced the relapse rate for those with SMI.45 Of note, short- and long-acting NRT can and should be combined with varenicline to increase the likelihood of smoking cessation.
Despite the removal of varenicline’s black box warning, many patients remain reticent to try this medication based on previous messaging by others or even their own experience. Patients’ concerns should be listened to, and they should be informed about recent evidence. Potential side effects should be anticipated before the drug’s initiation. Starting varenicline and initiating a cessation attempt at the same time may conflate the side effects of nicotine withdrawal with those of the medication. Therefore, varenicline may be started 1 month before a planned reduction in tobacco use or setting a quit date. The most common side effects of varenicline are gastrointestinal upset and insomnia. To combat these side effects, patients may take the medication with food and take their evening dose several hours before bedtime if sleep becomes problematic. In addition, for those who are sensitive to side effects, prescribers may consider scheduling a slower dose escalation than that provided in the varenicline starter pack or have their patients try varenicline at the lowest possible dose and keep them at this low dose until their next appointment.
What Are Some Common Myths and Challenges Faced by PCPs When Approaching Smoking Cessation?
Tobacco use is a leading contributor to the global burden of disease and to the 25- to 30-year mortality gap between people with SMI and the general population, thus a strategic focus on tobacco use during visits, when possible, has potential to make a large impact on both quality of life and longevity.5,46 PCPs may hear from their patients that smoking cigarettes reduces their anxiety. It may be helpful for clinicians to note that most of the perceived amelioration of psychiatric symptoms with cigarette use is short-lived at the outset of tobacco use, and the “anxiety relief” experienced when smoking a cigarette is largely due to blocking the symptoms of nicotine withdrawal that developed since an individual smoked their last cigarette. Long-term, cessation of tobacco use reduces symptoms of both depression and anxiety, with effect sizes equal to or larger than those of antidepressants.47 In the short-term, however, cessation and accompanying nicotine withdrawal may result in increased anxiety. It may be helpful to contrast this potential short-term increase with likely long-term improvement in psychiatric symptoms when talking to patients as they initiate a quit attempt.
Providers should take stock of their biases, for example, that people with SMI do not want to quit or that they will not quit successfully. Those with SMI do want to quit,48–50 although they may require more information about the health risks of continued smoking and the psychological benefits of smoking cessation.50,51 Rates of smoking cessation (with behavioral treatment alone) in SMI is ~4%, underscoring the importance of combining pharmacotherapy with behavioral treatments to be successful.24,26 Indeed, the EAGLES trial demonstrated higher rates of smoking cessation with pharmacotherapy among those with psychotic disorders than in the general population.24,36
Providers should ensure that medications are being used correctly, as many patients who report that an agent was ineffective may not have been using it properly (eg, chewing, not “parking,” nicotine gum; not using short-acting NRT frequently enough to get ahead of cravings; taking off the patch to smoke; taking varenicline sporadically). Additionally, if a medication was started at the same time as smoking was reduced or a cessation attempt was started, they may inaccurately attribute withdrawal symptoms to use of the medication.
Another common myth is that mentholated cigarettes are less harmful than those containing no flavors. This myth was created by an earlier aggressive marketing campaign of tobacco-producing companies that selectively targeted vulnerable populations (lower socioeconomic status, female black, etc). This myth is particularly dangerous not only because it undermines the effects of cigarette smoking but also because mentholated cigarettes have been found to be more dangerous than nonflavored cigarettes and perpetuate nicotine addiction. Research findings suggest that menthol might enhance the addictive potential of nicotine-containing products via several biological mechanisms including altering nicotinic receptors, masking the gustatory aversive sensations, acting as a conditioned cue due to its chemosensory properties, and ultimately by altering nicotine metabolism leading to higher bioavailability.52 These various menthol properties might explain the growing evidence that smokers of mentholated cigarettes are less likely to quit smoking or benefit from both behavioral and pharmacologic treatment interventions. These facts might be helpful to primary care providers in educating their patients about the additional risks of smoking mentholated cigarettes.
What Are the Benefits of Smoking Cessation?
Stopping cigarette smoking improves outcomes of both physical and mental health, even over the short-term, regardless of a person’s age or how long they have been smoking. According to a report by the Centers for Disease Control and Prevention,1 quitting smoking reduces the risk for cardiovascular diseases (eg, atherosclerosis, stroke, atrial fibrillation, heart failure, venous thromboembolism, and sudden cardiac death), respiratory diseases (eg, chronic obstructive pulmonary disease, infections such as pneumonia, bronchitis, and asthma), and cancer (eg, cancer of the lungs, esophagus, mouth, throat, and bladder). Moreover, a systematic review of studies revealed that smoking cessation was associated with reduced levels of depression, anxiety, and stress and an improved mood and quality of life compared to those who continued to smoke.47
Alternatively, drug concentration of psychiatric medications (eg, antipsychotics) in blood can be decreased with cigarette smoking. Thus, smokers often require inappropriately higher doses of psychiatric medications than nonsmokers. Smoking cessation may often lead to a reduction of medication dosages thus decreasing the risk of drug-related side effects. Since patients with SMI, on average, spend more of their income on tobacco, smoking cessation can reduce financial stress, improve overall mental health and well-being, and boost emotional and psychological relationships. Moreover, quitting cigarette smoking may also protect family members, coworkers, and friends from the health risks of second-hand smoke.
How Can Cutting Back on Cigarette Smoking Affect Drug Levels of Coadministered Drugs?
Tobacco smoking affects the serum levels of many coadministered drugs due to induction of cytochrome P450 (CYP) hepatic enzymes (especially CYP1A2) by polycyclic aromatic hydrocarbons and non-nicotinic chemical constituents, resulting in more rapid metabolism and clearance of certain drugs.53,54 Of note, nicotine itself has no effect on the CYP enzymes, and nicotine replacement formulations do not affect the serum levels of coadministered medications like tobacco smoking does. Some drugs affected by the CYP1A2 isoenzyme system include psychiatric medications (eg, clozapine, olanzapine, imipramine, diazepam, ramelteon), antihypertensive drugs (eg, propranolol, verapamil), opioid analgesics (eg, propoxyphene), anticoagulants (warfarin), and drugs used to treat asthma and other respiratory problems (eg, theophylline).53–56 Another effect of tobacco smoking includes reduction of cutaneous and subcutaneous blood flow, thus slowing the absorption of injectable drugs, such as insulin. Drug interactions with tobacco smoking can also lead to activation of thrombosis and faster clearance of heparin. Smoking cessation normalizes levels of hepatic CYP enzymes and reverses other smoking-induced effects, significantly increasing plasma drug concentrations (by up to 40%).55
When PCPs discuss options for smoking cessation with their patients, they should consider how different interventions may affect levels of coadministered drugs. Effective and timely monitoring and management of coadministered drugs remains vital to preventing serious adverse consequences associated with higher serum levels, which are especially important for those medications with a narrow therapeutic index and that may lead to toxicity (eg, with use of clozapine, olanzapine, warfarin, and theophylline).56,57 Dose adjustments should be based on both clinical and laboratory findings (eg, plasma serum levels). In addition, more frequent in-person or telephonic/telehealth check-ins may be needed until a safe and effective dose has been determined. While a detailed description of recommended dose adjustments for each coadministered drug is beyond the scope of this article, a reference table has been provided by the Smoking Cessation and Leadership Center, University of California and San Francisco.58
Additional attention should be given to smoking cessation in the elderly, given their susceptibility to drug side effects associated with age-related pharmacodynamics and pharmacokinetic changes and a greater incidence of comorbidities. Patients with psychiatric disorders, especially those with SMI, require extra attention to their higher prevalence of coadministered antipsychotics and other psychotropics. Health care providers should also educate their patients (and their patients’ family members) about the signs and symptoms of drug toxicity from coadministered drugs and create a safety plan that includes placement in a higher level of care to ensure their safety. This is especially important in the elderly and in those with SMI whose age- or disease-related cognitive impairment often impairs their ability to observe and report medication side effects that can accompany smoking cessation.56
Case Follow-Up
After 6 months, Mr A continued to take varenicline 1 mg twice/day with no side effects and was able to decrease his tobacco use to 4 cigarettes/day. He was pleased to be saving money and was able to reduce the dose of his antipsychotic medication after cutting down the number of cigarettes. In addition to varenicline, he also used nicotine gum as needed for breakthrough cravings and was hopeful about the possibility of quitting cigarettes entirely soon. His mental health condition remained stable, and he had begun to explore job options.
CONCLUSION
Tobacco use remains a leading cause of death globally and should be routinely addressed in the primary care setting.1,3 In particular, patients with SMI have elevated rates of morbidity and mortality from smoking-related diseases such as cancer, heart disease, diabetes, and lung disorders; thus, smoking cessation should be a high priority for PCPs who are providing long-term care to patients with mental illness. Evidence-based behavioral and pharmacotherapeutic interventions (such as NRT, bupropion, varenicline) have been shown to enhance abstinence rates and result in health benefits among patients with SMI. Our case example highlights that PCPs are well-positioned to help patients quit smoking by facilitating conversations about the cessation process, including withdrawal symptoms, medication use, and behavioral treatments, and employing tailored interventions.
Submitted: February 17, 2021; accepted September 24, 2021.
Published online: May 17, 2022.
Relevant financial relationships: None.
Funding/support: None.
Clinical Points
- Patients with severe mental illness have elevated rates of morbidity and mortality from smoking-related diseases such as cancer, heart disease, diabetes, and lung disorders.
- While behavioral health interventions and use of medications can facilitate smoking cessation, pharmacotherapy and counseling are most successful when used in tandem.
- Smoking cessation should be a high priority for primary care physicians who are providing long-term care to patients with mental illness.
References (58)

- United States Public Health Service Office of the Surgeon General; National Center for Chronic Disease Prevention and Health Promotion (US) Office on Smoking and Health. Smoking Cessation: A Report of the Surgeon General. Washington, DC: US Department of Health and Human Services; 2020:439–486.
- Goldenberg M, Danovitch I, IsHak WW. Quality of life and smoking. Am J Addict. 2014;23(6):540–562. PubMed CrossRef
- National Center for Chronic Disease Prevention and Health Promotion (US) Office on Smoking and Health. The Health Consequences of Smoking - 50 Years of Progress: A Report of the Surgeon General. Atlanta (GA). US: Centers for Disease Control and Prevention; 2014.
- Samet JM. Tobacco smoking: the leading cause of preventable disease worldwide. Thorac Surg Clin. 2013;23(2):103–112. PubMed CrossRef
- Olfson M, Gerhard T, Huang C, et al. Premature mortality among adults with schizophrenia in the United States. JAMA Psychiatry. 2015;72(12):1172–1181. PubMed CrossRef
- National Cancer Institute. “How Much Will You Save?” Smokefree.gov website. Accessed January 18, 2021. https://smokefree.gov/quit-smoking/why-you-should-quit/how-much-will-you-save
- Lindson N, Thompson TP, Ferrey A, et al. Motivational interviewing for smoking cessation. Cochrane Database Syst Rev. 2019;7(7):CD006936. PubMed
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders: DSM 5. Washington: American Psychiatric Association; 2013.
- Hughes JR. Effects of abstinence from tobacco: valid symptoms and time course. Nicotine Tob Res. 2007;9(3):315–327. PubMed CrossRef
- Covey LS, Glassman AH, Stetner F. Depression and depressive symptoms in smoking cessation. Compr Psychiatry. 1990;31(4):350–354. PubMed CrossRef
- Covey LS, Glassman AH, Stetner F. Major depression following smoking cessation. Am J Psychiatry. 1997;154(2):263–265. PubMed CrossRef
- Breslau N, Kilbey MM, Andreski P. Nicotine withdrawal symptoms and psychiatric disorders: findings from an epidemiologic study of young adults. Am J Psychiatry. 1992;149(4):464–469. PubMed CrossRef
- Flannery JS, Riedel MC, Poudel R, et al. Habenular and striatal activity during performance feedback are differentially linked with state-like and trait-like aspects of tobacco use disorder. Sci Adv. 2019;5(10):eaax2084. PubMed CrossRef
- Koob GF, Volkow ND. Neurocircuitry of addiction. Neuropsychopharmacology. 2010;35(1):217–238. PubMed CrossRef
- McLaughlin I, Dani JA, De Biasi M. Nicotine withdrawal. Curr Top Behav Neurosci. 2015;24:99–123. PubMed CrossRef
- Sweitzer MM, Geier CF, Joel DL, et al. Dissociated effects of anticipating smoking versus monetary reward in the caudate as a function of smoking abstinence. Biol Psychiatry. 2014;76(9):681–688. PubMed CrossRef
- Wilson SJ, Delgado MR, McKee SA, et al. Weak ventral striatal responses to monetary outcomes predict an unwillingness to resist cigarette smoking. Cogn Affect Behav Neurosci. 2014;14(4):1196–1207. PubMed CrossRef
- George TP, Vessicchio JC, Termine A, et al. Effects of smoking abstinence on visuospatial working memory function in schizophrenia. Neuropsychopharmacology. 2002;26(1):75–85. PubMed CrossRef
- Piasecki TM, Jorenby DE, Smith SS, et al. Smoking withdrawal dynamics, II: improved tests of withdrawal-relapse relations. J Abnorm Psychol. 2003;112(1):14–27. PubMed CrossRef
- Smith PH, Homish GG, Giovino GA, et al. Cigarette smoking and mental illness: a study of nicotine withdrawal. Am J Public Health. 2014;104(2):e127–e133. PubMed CrossRef
- Rigotti NA. Strategies to help a smoker who is struggling to quit. JAMA. 2012;308(15):1573–1580. PubMed CrossRef
- Maglione MA, Maher AR, Ewing B, et al. Efficacy of mindfulness meditation for smoking cessation: a systematic review and meta-analysis. Addict Behav. 2017;69:27–34. PubMed CrossRef
- Wong SY, Zhang DX, Li CC, et al. Comparing the effects of mindfulness-based cognitive therapy and sleep psychoeducation with exercise on chronic insomnia: A randomied controlled trial. Psychother Psychosom. 2017;86(4):241–253. PubMed CrossRef
- Evins AE, Benowitz NL, West R, et al. Neuropsychiatric safety and efficacy of varenicline, bupropion, and nicotine patch in smokers with psychotic, anxiety, and mood disorders in the EAGLES trial. J Clin Psychopharmacol. 2019;39(2):108–116. PubMed CrossRef
- Fiore MC, Jaén CR, Baker TB, et al. Treating Tobacco Use and Dependence: 2008 Update. Clinical Practice Guideline. Rockville, MD: US Department of Health and Human Services. Public Health Service. May 2008.
- Tsoi DT, Porwal M, Webster AC. Interventions for smoking cessation and reduction in individuals with schizophrenia. Cochrane Database Syst Rev. 2013;2013(2):CD007253. PubMed CrossRef
- Roberts NJ, Kerr SM, Smith SM. Behavioral interventions associated with smoking cessation in the treatment of tobacco use. Health Serv Insights. 2013;6:79–85. PubMed CrossRef
- DiClemente CC, Delahanty JC, Kofeldt MG, et al. Stage movement following a 5A’s intervention in tobacco dependent individuals with serious mental illness (SMI). Addict Behav. 2011;36(3):261–264. PubMed CrossRef
- Cather C, Pachas GN, Cieslak KM, et al. Achieving smoking cessation in individuals with schizophrenia: special considerations. CNS Drugs. 2017;31(6):471–481. PubMed CrossRef
- Hartmann-Boyce J, Chepkin SC, Ye W, et al. Nicotine replacement therapy versus control for smoking cessation. Cochrane Database Syst Rev. 2018;5:CD000146. PubMed CrossRef
- Carpenter MJ, Jardin BF, Burris JL, et al. Clinical strategies to enhance the efficacy of nicotine replacement therapy for smoking cessation: a review of the literature. Drugs. 2013;73(5):407–426. PubMed CrossRef
- Heil SH, Linares Scott T, Higgins ST. An overview of principles of effective treatment of substance use disorders and their potential application to pregnant cigarette smokers. Drug Alcohol Depend. 2009;104(suppl 1):S106–S114. PubMed CrossRef
- de Leon J, Diaz FJ, Rogers T, et al. Initiation of daily smoking and nicotine dependence in schizophrenia and mood disorders. Schizophr Res. 2002;56(1–2):47–54. PubMed CrossRef
- Williams JM, Gandhi KK, Lu SE, et al. Higher nicotine levels in schizophrenia compared with controls after smoking a single cigarette. Nicotine Tob Res. 2010;12(8):855–859. PubMed CrossRef
- Williams JM, Ziedonis DM, Abanyie F, et al. Increased nicotine and cotinine levels in smokers with schizophrenia and schizoaffective disorder is not a metabolic effect. Schizophr Res. 2005;79(2-3):323–335. PubMed CrossRef
- Anthenelli RM, Benowitz NL, West R, et al. Neuropsychiatric safety and efficacy of varenicline, bupropion, and nicotine patch in smokers with and without psychiatric disorders (EAGLES): a double-blind, randomised, placebo-controlled clinical trial. Lancet. 2016;387(10037):2507–2520. PubMed CrossRef
- Piper ME, Smith SS, Schlam TR, et al. A randomized placebo-controlled clinical trial of 5 smoking cessation pharmacotherapies. Arch Gen Psychiatry. 2009;66(11):1253–1262. PubMed CrossRef
- Smith SS, McCarthy DE, Japuntich SJ, et al. Comparative effectiveness of 5 smoking cessation pharmacotherapies in primary care clinics. Arch Intern Med. 2009;169(22):2148–2155. PubMed CrossRef
- Schnoll RA, Goelz PM, Veluz-Wilkins A, et al. Long-term nicotine replacement therapy: a randomized clinical trial. JAMA Intern Med. 2015;175(4):504–511. PubMed CrossRef
- Mills EJ, Wu P, Lockhart I, et al. Adverse events associated with nicotine replacement therapy (NRT) for smoking cessation: a systematic review and meta-analysis of one hundred and twenty studies involving 177,390 individuals. Tob Induc Dis. 2010;8(1):8. PubMed CrossRef
- Hajek P, Phillips-Waller A, Przulj D, et al. A randomized trial of e-cigarettes versus nicotine-replacement therapy. N Engl J Med. 2019;380(7):629–637. PubMed CrossRef
- Pearsall R, Smith DJ, Geddes JR. Pharmacological and behavioural interventions to promote smoking cessation in adults with schizophrenia and bipolar disorders: a systematic review and meta-analysis of randomised trials. BMJ Open. 2019;9(11):e027389. PubMed CrossRef
- Simon JA, Duncan C, Carmody TP, et al. Bupropion for smoking cessation: a randomized trial. Arch Intern Med. 2004;164(16):1797–1803. PubMed CrossRef
- Evins AE, West R, Benowitz NL, et al. Efficacy and safety of pharmacotherapeutic smoking cessation aids in schizophrenia spectrum disorders: Subgroup analysis of EAGLES. Psychiatr Serv. 2021;72(1):7–15. PubMed CrossRef
- Evins AE, Hoeppner SS, Schoenfeld DA, et al. Maintenance pharmacotherapy normalizes the relapse curve in recently abstinent tobacco smokers with schizophrenia and bipolar disorder. Schizophr Res. 2017;183:124–129. PubMed CrossRef
- Parks J, Svendsen D, Singer P, et al. Morbidity and Mortality in People With Serious Mental Illness. Alexandria, VA: National Association of State Mental Health Program Directors (NASMHPD) Medical Directors Council. 2006;25(4):1–87.
- Taylor G, McNeill A, Girling A, et al. Change in mental health after smoking cessation: systematic review and meta-analysis. BMJ. 2014;348(feb13 1):g1151. PubMed CrossRef
- Siru R, Hulse GK, Tait RJ. Assessing motivation to quit smoking in people with mental illness: a review. Addiction. 2009;104(5):719–733. PubMed CrossRef
- Addington J, el-Guebaly N, Addington D, et al. Readiness to stop smoking in schizophrenia. Can J Psychiatry. 1997;42(1):49–52. PubMed CrossRef
- Kelly DL, Raley HG, Lo S, et al. Perception of smoking risks and motivation to quit among nontreatment-seeking smokers with and without schizophrenia. Schizophr Bull. 2012;38(3):543–551. PubMed CrossRef
- Tidey JW, Rohsenow DJ. Smoking expectancies and intention to quit in smokers with schizophrenia, schizoaffective disorder and non-psychiatric controls. Schizophr Res. 2009;115(2–3):310–316. PubMed CrossRef
- Wickham RJ. The biological impact of menthol on tobacco dependence. Nicotine Tob Res. 2020;22(10):1676–1684. PubMed CrossRef
- Kroon LA. Drug interactions with smoking. Am J Health Syst Pharm. 2007;64(18):1917–1921. PubMed CrossRef
- Li H, Shi Q. Drugs and diseases interacting with cigarette smoking in US prescription drug labelling. Clin Pharmacokinet. 2015;54(5):493–501. PubMed CrossRef
- Faber MS, Fuhr U. Time response of cytochrome P450 1A2 activity on cessation of heavy smoking. Clin Pharmacol Ther. 2004;76(2):178–184. PubMed CrossRef
- Schaffer SD, Yoon S, Zadezensky I. A review of smoking cessation: potentially risky effects on prescribed medications. J Clin Nurs. 2009;18(11):1533–1540. PubMed CrossRef
- Lowe EJ, Ackman ML. Impact of tobacco smoking cessation on stable clozapine or olanzapine treatment. Ann Pharmacother. 2010;44(4):727–732. PubMed CrossRef
- Smoking Cessation Leadership Center. University of California San Francisco website. Accessed January 19, 2021. https://smokingcessationleadership.ucsf.edu/factsheets/drug-interactions-tobacco-smoke-rx-change-2019
Please sign in or purchase this PDF for $40.
Save
Cite