Find more articles on this and other psychiatry and CNS topics:
The Journal of Clinical Psychiatry
The Primary Care Companion for CNS Disorders
CASE CONFERENCE
The Banner Alzheimer’s Institute Case Conference is a weekly event in which physicians and staff discuss challenging and/or teaching cases of patients seen at the Institute’s Stead Family Memory Clinic. These conferences are attended by a multidisciplinary group that includes Banner Alzheimer’s Institute dementia specialists, community physicians (internal medicine, family medicine, and radiology), physician assistants, social workers, nurses, medical students, residents, and fellows.
BANNER ALZHEIMER’ S INSTITUTE
The Banner Alzheimer’s Institute located in Phoenix, Arizona, has an unusually ambitious mission: to end Alzheimer’s disease without losing a generation, set a new standard of care for patients and families, and forge a model of collaboration in biomedical research. The Institute provides high-level care and treatment for patients affected by Alzheimer’s disease, dementia, and related disorders. In addition, the Institute offers extensive support services for families and many unique and rewarding research opportunities.
Prim Care Companion CNS Disord 2013;15(5):doi:10.4088/PCC.13alz01595
© Copyright 2013 Physicians Postgraduate Press, Inc.
Received: October 11, 2013; accepted October 11, 2013.
Published online: October 31, 2013.
AUTHORS
Anna D. Burke, MD, is a geriatric psychiatrist and dementia specialist at the Stead Family Memory Clinic of Banner Alzheimer’s Institute.
Roy Yaari, MD, MAS, a neurologist, is associate director of the Stead Family Memory Clinic of Banner Alzheimer’s Institute and a clinical professor of neurology at the College of Medicine, University of Arizona, Phoenix.
Pierre N. Tariot, MD, a geriatric psychiatrist, is director of Banner Alzheimer’s Institute and a research professor of psychiatry at the College of Medicine, University of Arizona, Phoenix.
Adam S. Fleisher, MD, MAS, is associate director of Brain Imaging at the Banner Alzheimer’s Institute, a neurologist at the Stead Family Memory Clinic, and an associate professor in the Department of Neurosciences at the University of California, San Diego.
Geri R. Hall, PhD, ARNP, GCNS, FAAN, is a gerontology clinical nurse specialist at Banner Alzheimer’s Institute and an adjunct clinical professor at the College of Nursing, University of Arizona, Tucson.
Helle Brand, PA, is a physician assistant at the Stead Family Memory Clinic of Banner Alzheimer’s Institute.
Corresponding author: Anna D. Burke, MD, Banner Alzheimer’s Institute, 901 E. Willetta St, Phoenix, AZ 85006 ([email protected]).
CME Background
Original material is selected for credit designation based on an assessment of the educational needs of CME participants, with the purpose of providing readers with a curriculum of CME activities on a variety of topics from volume to volume. This special series of case reports about dementia was deemed valuable for educational purposes by the Publisher, Editor in Chief, and CME Institute Staff. Activities are planned using a process that links identified needs with desired results.
To obtain credit, read the material and go to http://www.cmeinstitute.com/activities/journal.asp to complete the Posttest and Evaluation online.
CME Objective
After studying this case, you should be able to:
- Provide management strategies for elderly patients with dementia who have inappropriate sexual behaviors
Accreditation Statement
The CME Institute of Physicians Postgraduate Press, Inc., is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians.
Credit Designation
The CME Institute of Physicians Postgraduate Press, Inc., designates this journal-based CME activity for a maximum of 1.0 AMA PRA Category 1 Creditâ„¢. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
Note: The American Academy of Physician Assistants (AAPA) accepts certificates of participation for educational activities certified for AMA PRA Category 1 Creditâ„¢ from organizations accredited by ACCME or a recognized state medical society. Physician assistants may receive a maximum of 1.0 hour of Category I credit for completing this program.
Date of Original Release/Review
This educational activity is eligible for AMA PRA Category 1 Creditâ„¢ through October 31, 2016. The latest review of this material was October 2013.
Financial Disclosure
All individuals in a position to influence the content of this activity were asked to complete a statement regarding all relevant personal financial relationships between themselves or their spouse/partner and any commercial interest. The CME Institute has resolved any conflicts of interest that were identified. In the past year, Larry Culpepper, MD, MPH, Editor in Chief, has been a consultant for AstraZeneca, Forest, Janssen, Lundbeck, Merck, Pfizer, and Takeda and has been a member of the speakers/advisory board for Merck. No member of the CME Institute staff reported any relevant personal financial relationships. Faculty financial disclosure appears at the end of the article.
HISTORY OF PRESENTING ILLNESS
Mr A is a 73-year-old married man who initially presented to the Stead Family Memory Clinic at Banner Alzheimer’s Institute in 2010 with his wife, who supplemented the clinical history, for evaluation of gradually progressing cognitive impairment of approximately 2 years’ duration. In early 1996, he was diagnosed with and treated for Parkinson’s disease. However, it was not until 2008 when Mr A and his wife reported an insidious onset of short-term memory difficulties, including repetition of stories and fluctuating periods of confusion regarding events and appointments. Mr A’s neurologist then started donepezil, which was stopped for unclear reasons. The rivastigmine transdermal patch was subsequently initiated but was stopped approximately a year prior to his appointment at Banner Alzheimer’s Institute due to “worsening of parkinsonian symptoms.” After stopping the rivastigmine patch, a rapid decline in cognition was observed. Mr A became increasingly confused and rapidly forgetful. He would at times stand with the TV remote in his hand and be unable to use it. He would open the dishwasher to put away the dishes and then leave it open and walk away, forgetting to put any of the dishes in it. He was unable to focus and complete tasks. His wife noticed significant increases in repetition of questions and conversations, difficulty reading and writing, and misplacing items around the house. As a result, oral rivastigmine was restarted after “several weeks,” and cognitive symptoms significantly improved, but not back to his baseline level prior to discontinuation of the rivastigmine patch.
At the time of the visit to Banner Alzheimer’s Institute, Mr A, who had now been on oral rivastigmine for several months, reported continued worsening of short-term memory. He was frequently disoriented to time and date. He was repetitive in conversations. He had difficulty maintaining a calendar and remembering appointments. Difficulty concentrating on tasks was also noted. Mr A was having more difficulty with reading and writing and misplacing items. He was able to remember people’s names and recognize familiar faces.
Most personal activities of daily living were maintained, but Mr A was experiencing greater difficulty performing instrumental activities of daily living. Although his Parkinson’s disease had resulted in changes in fine motor movements, making it difficult for him to fasten buttons and tie shoelaces, Mr A could now no longer coordinate tasks such as putting on pants or T-shirts. For example, there was one incident in which Mr A put his clothes on backward. He also required prompting to remember to bathe and brush his teeth. Mr A could no longer shop by himself, manage finances, or calculate tips and handle cash.
Mr A continued to drive, although he had self-limited his driving due to what he perceived as Parkinson’s -related decreased reflexes. Although there were no motor vehicle accidents on the road or instances of getting lost, there were several episodes of hitting the house with the car and with a golf cart. No confusion while driving was reported by Mr A’s spouse.
Mr A’s appetite was poor, and he was losing weight. He also struggled with insomnia and was frequently using clonazepam as a sleep aid. No urinary or fecal incontinence had been reported.
Mr A was described lovingly by family as always being outgoing and having a great sense of humor. Over the past several years, he had become more withdrawn and apathetic. He reported a subjective sense of emotional lability with tearful episodes. His primary care physician subsequently started him on sertraline.
Other personality changes included occasional inappropriate comments or behaviors, including sexually inappropriate comments. Although Mr A had been impotent since 1991, he had recently experienced an increase in libido, requesting sexual activity 3 to 4 times per week. His wife found that she was extremely uncomfortable with her husband in a sexual manner and had told him that she no longer wished to engage in sexual activity with him. Consequently, he became angry and obsessive about sex. She stated that he would ask her for sexual relations and repeatedly question her as to why she did not wish to have relations with him. He continued to perseverate on this topic and talk with people and family members about sex in public settings, which was very uncharacteristic of him. He would also intermittently make self-deprecating statements and suggest that he would be better off committing suicide since he felt that he was “no longer a man.”
Mr A admitted to vague psychotic features such as “feeling” the presence of people around him, which was quite distressing to him. He also experienced visual hallucinations of bugs and occasional visions of “blurred people,” mainly occurring during the evening or early morning hours with a tendency to be precipitated by darkness.
Sexual disinhibition occurs in 2%-17% of patients with dementia (Burns et al, 1990). Sexually inappropriate behaviors may range from inappropriate comments and solicitations and inappropriate sexual activity (eg, masturbation in public places, exposing breasts or genitalia in public) to aggression related to sexual impulsivity. These behaviors occur with equal frequency in males and females (Burns et al, 1990).
Series and Degano (2005) postulated potential causes for development of sexually inappropriate behaviors. These causes include disease-related factors such as neurodegeneration of the frontal lobes and development of a Kl×¼ver-Bucy syndrome, forgetting normal etiquette, development of sexually oriented delusions or misidentifications, and sensory impairments. Social factors promoting inappropriate sexual activity include lack of usual sexual partners, lack of privacy, an understimulating environment, misinterpretation of cues such as those seen on TV or in opposite-gender caregivers, and an unfamiliar environment. Psychological factors such as premorbid patterns of sexual activity and interest, as well as changes in mood states (depression/mania) will also affect sexual interest in dementia. Many medications and substances (eg, alcohol and benzodiazepines) can produce behavioral and sexual disinhibition. Treatment of parkinsonian symptoms with levodopa has also been shown to be related to development of manic-type symptoms and hypersexuality (Series and Degano, 2005).
PAST MEDICAL HISTORY
Mr A reported a past medical history significant for hypothyroidism, hypercholesterolemia, insomnia, coronary artery disease with 3 angioplasties and a double bypass surgery, bilateral carotid endarterectomies, back surgery for sciatica, diagnosis of Parkinson’s disease in 1996, and transient ischemic attacks.
ALLERGIES
No known drug allergies.
MEDICATIONS
Mr A’s medications included levothyroxine 125 μg daily, atorvastatin 40 mg daily, clonazepam 0.5 mg nightly, carbidopa/levodopa 25/100 mg per morning and carbidopa/levodopa controlled release 50/200 mg twice daily, rivastigmine 6 mg twice daily orally, aspirin 81 mg daily, vitamin D, and venlafaxine extended release 150 mg daily. Over the course of the past 2 weeks, Mr A had been switched to venlafaxine because sertraline 150 mg daily had little to no effect.
SOCIAL HISTORY
Mr A had 12 years of education and worked as a computer specialist for over 30 years. He was currently retired and living with his spouse independently in their home.
SUBSTANCE ABUSE HISTORY
No known history of alcohol or illicit drug use. Mr A reported that he smoked cigarettes, 2 packs per day, for approximately 35 years before quitting in the 1980s.
FAMILY HISTORY
Mr A’s mother had “dementia,” and his brother had a history of stroke. He denied any history of Parkinson’s disease or mental illness in his family.
REVIEW OF SYSTEMS
A complete review of systems was positive for diminished appetite and sense of smell and mild weight loss, as well as insomnia and low energy levels. Visual and auditory impairment was noted. His speech had become more “mumbling,” slurred, and hypophonic. Intermittent dysphagia was reported. Mr A’s review of systems was also positive for constipation, urinary frequency and urgency, hip and neck pain, resting tremor, limb weakness, and difficulty with balance. Hallucinations and emotional lability, as described above, were also noted.
PHYSICAL EXAMINATION
Mr A was a well-nourished, well-developed man displaying significant psychomotor retardation. Ophthalmic examination revealed decreased clarity of the lenses of both eyes. Cardiac examination revealed a mild systolic murmur; rate and rhythm were normal. A slight kyphosis was present.
NEUROLOGIC EXAMINATION
Mr A’s neurologic examination revealed bilateral decreased hearing to finger rub, hypomimia, and decreased blink. Speech was hypophonic with decreased spontaneous verbal output. Cerebellar testing was significant for mild right dysmetria. Deep tendon reflexes were hypoactive but symmetric throughout. Gait was slow, shuffling, and unsteady with turn en bloc. Muscle tone was rigid and increased in both upper extremities with bilateral cogwheeling present. A low-frequency, high-amplitude resting tremor in both hands, right greater than left, was noted. Dyskinetic movements of the neck, tongue, and legs were present.
MENTAL STATUS EXAMINATION
Mr A was a very well-groomed elderly white man appearing to be in no acute distress. He displayed appropriate eye contact with decrease in blink. He displayed psychomotor retardation. Mr A’s mood was “good.” His affect was, for the most part, constricted, although he displayed occasional mood lability with tearfulness and excessive reactivity to negative stimuli. Thought process was coherent, logical, and goal directed. There was no evidence of any paranoid, suicidal, or homicidal ideations. No auditory hallucinations were noted at the time of the visit. Mr A endorsed visual hallucinations. Judgment and insight were impaired. Fund of knowledge was normal for age and education level. Mr A was oriented to time, place, and person. Attention and concentration were limited.
LABORATORIES/RADIOLOGY
No laboratory studies or neuroimaging results were available at the time of the visit.
Dementia is defined as a cluster of multiple cognitive deficits that include memory impairment and at least 1 of the following cognitive disturbances: aphasia, apraxia, agnosia, or a disturbance in executive functioning. The cognitive deficits must be sufficiently severe to cause impairment in social or occupational functioning and must represent a decline from a previously higher level of functioning. A diagnosis of dementia should not be made if the cognitive deficits occur exclusively during the course of a delirium.
On the basis of the information so far, do you think a dementia is present?
Your Colleagues Who Attended the Banner Alzheimer’s Institute Case Conference Answered as Follows:
| A. Yes | 100% |
| B. No | 0% |
| C. Not enough information | 0% |
All of the conference attendees felt that Mr A met criteria for a dementia due to the presence of cognitive impairments affecting his functional abilities.A Mini-Mental State Examination (MMSE) score generally correlates with disease severity. Scores ≤ 9 points can indicate severe dementia, scores between 10-20 points can indicate moderate dementia, and scores > 20 points can indicate mild dementia (Mungas, 1991). Although MMSE scores must be interpreted in light of both the patient’s age and education, education is the primary demographic factor that affects scores. Therefore, whereas a cutoff ≤ 23 is widely used in distinguishing between normal and abnormal performance, this cutoff may have less predictive ability in poorly educated individuals (Folstein et al, 1975).
On the basis of the information so far, what would you expect the MMSE score to be?
Your Colleagues Who Attended the Banner Alzheimer’s Institute Case Conference Answered as Follows:
| A. 26-30 | 34% |
| B. 21-25 | 33% |
| C. 16-20 | 33% |
| D. 11-15 | 0% |
| E. < 11 | 0% |
Mr A’s MMSE score was 26/30. Mr A’s sentence and pentagon drawing from the MMSE are shown in Figure 1. The Montreal Cognitive Assessment (MoCA) is a 30-point test that assesses several cognitive domains. Because it is more challenging than the Mini-Mental State Examination, it has greater sensitivity for mild cognitive impairment and early stages of dementia. With a cutoff score < 26, the sensitivity for detecting mild cognitive impairment (N = 94) was 90% and the specificity was 87% (Nasreddine et al, 2005). Research has demonstrated that MoCA scores are highly correlated with education. It is recommended that education be taken into account when interpreting MoCA performance, but there are no formal specific cutoff scores for lower education at this time (Johns et al, 2008, a, b). This test is available online at http://mocatest.org/.
The Montreal Cognitive Assessment (MoCA) is a 30-point test that assesses several cognitive domains. Because it is more challenging than the Mini-Mental State Examination, it has greater sensitivity for mild cognitive impairment and early stages of dementia. With a cutoff score < 26, the sensitivity for detecting mild cognitive impairment (N = 94) was 90% and the specificity was 87% (Nasreddine et al, 2005). Research has demonstrated that MoCA scores are highly correlated with education. It is recommended that education be taken into account when interpreting MoCA performance, but there are no formal specific cutoff scores for lower education at this time (Johns et al, 2008, a, b). This test is available online at http://mocatest.org/.
On the basis of the information so far, what would you expect the Montreal Cognitive Assessment (MoCA) score to be?
Your Colleagues Who Attended the Banner Alzheimer’s Institute Case Conference Answered as Follows:
| A. 26-30 | 0% |
| B. 21-25 | 60% |
| C. 16-20 | 40% |
| D. 11-15 | 0% |
| E. < 11 | 0% |
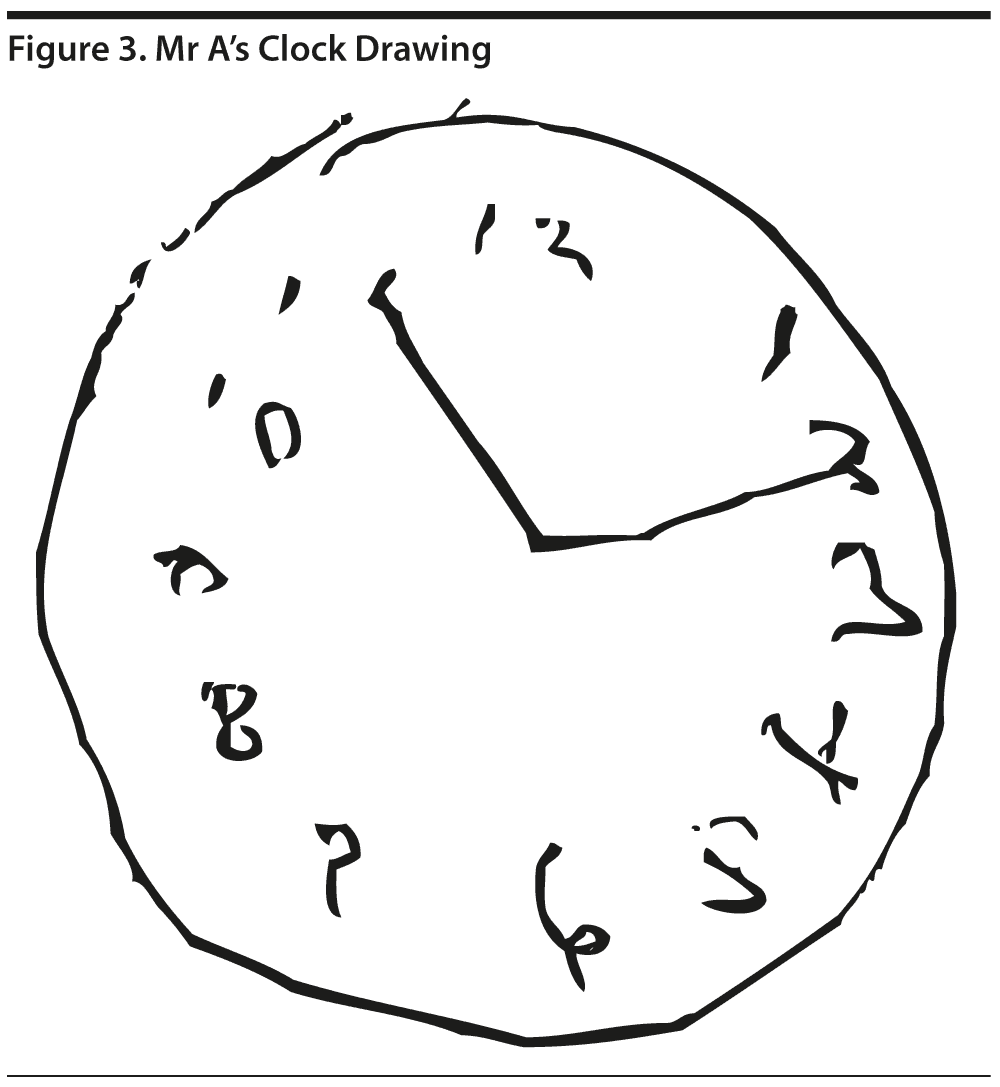
Mr A’s MoCA score was 23/30. Mr A’s MoCA results and clock drawing are shown in Figures 2 and 3, respectively.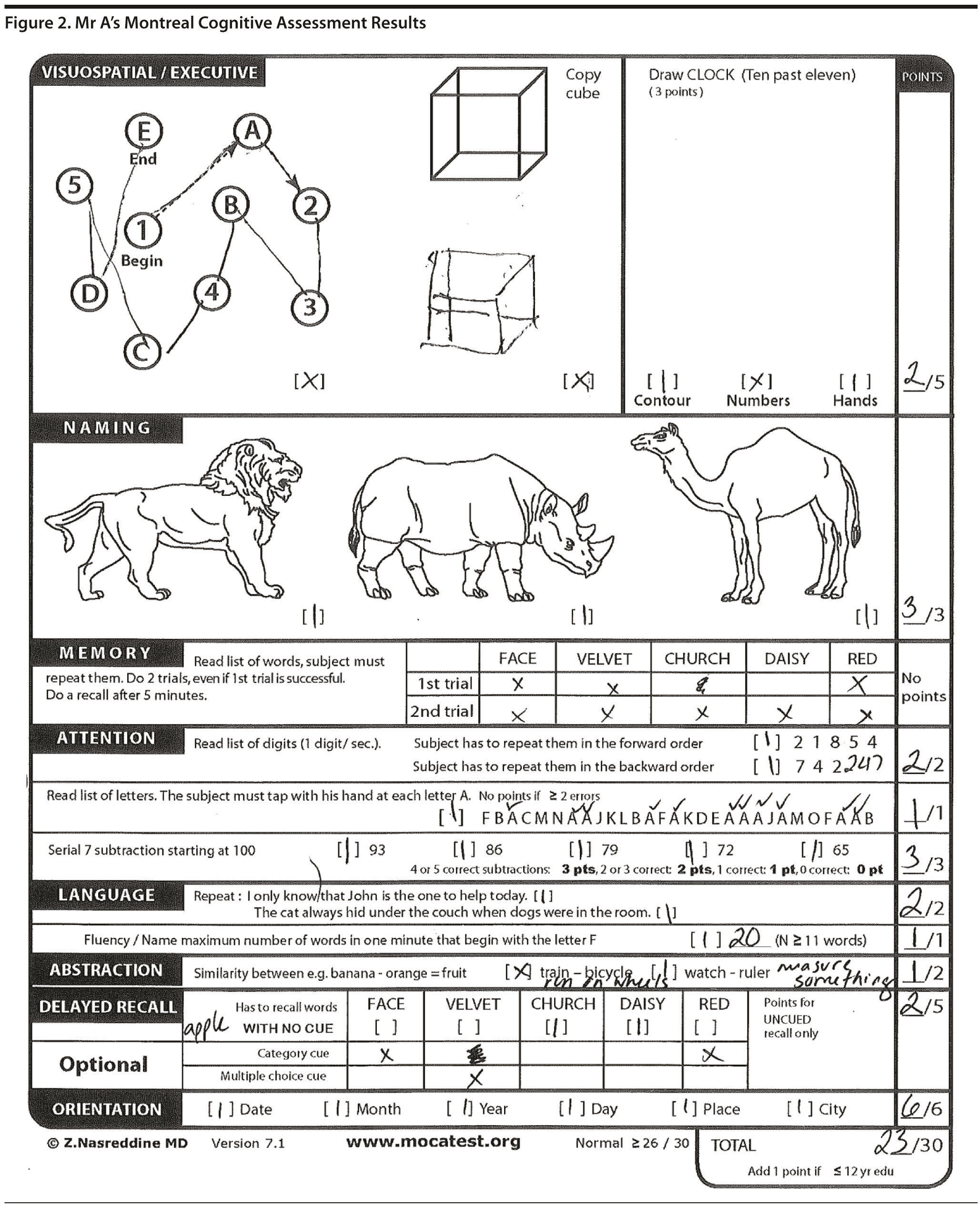
On the basis of the information so far, what is the most likely primary diagnosis?
Your Colleagues Who Attended the Banner Alzheimer’s Institute Case Conference Answered as Follows:
| A. Dementia with Lewy bodies | 15% |
| B. Parkinson’s dementia | 50% |
| C. Frontotemporal dementia syndrome | 0% |
| D. Alzheimer’s dementia | 25% |
| E. Vascular dementia | 10% |
| F. Dementia not otherwise specified | 0% |
The majority of those attending the case conference felt that Mr A’s cognitive impairment was related to Parkinson’s disease and met criteria for a Parkinson’s dementia. Some argued that, due to the pattern of deficits on screening tests, which appeared to be mainly encompassing short-term memory abilities, and due to the prevalence of the disorder, Alzheimer’s dementia was a potential diagnosis.The diagnosis of Parkinson’s disease with dementia versus dementia with Lewy bodies is based on the clinical evaluation of the timing of onset of motor and cognitive symptoms. Cognitive symptoms tend to appear within the first year of onset of parkinsonian symptoms in dementia with Lewy bodies, but do not emerge until several years after onset of parkinsonian symptoms in Parkinson’s disease with dementia. Both syndromes share many neurobiological similarities including the presence of Lewy bodies, which appears to be more diffuse in dementia with Lewy bodies. Mixed Alzheimer’s disease pathology with deposition of β-amyloid protein is also more common and more closely related to cognitive impairment in dementia with Lewy bodies than Parkinson’s disease dementia, possibly contributing to earlier dementia at onset. Executive dysfunction appears to be less prominent in Parkinson’s disease dementia and may relate to lesser loss of frontohippocampal projections in Parkinson’s disease dementia. Abundant Lewy body pathology in temporal cortex in dementia with Lewy bodies is associated with greater prevalence of visual hallucinations and delusions.
What should the next step be?
Your Colleagues Who Attended the Banner Alzheimer’s Institute Case Conference Answered as Follows:
| A. Neuropsychological testing | 0% |
| B. Laboratories (CBC, CMP, TSH, vitamin B12 level) | 0% |
| C. Structural brain scan | 0% |
| D. Amyloid PET scan | 0% |
| E. FDG-PET scan | 0% |
| F. B and C | 0% |
| G. A, B, C | 100% |
Those attending the case conference agreed that the most appropriate next step would be ordering structural brain imaging to rule out any intracranial pathology and laboratory studies to rule out metabolic and infectious causes. Attendees also suggested that a neuropsychological battery should be performed to help clarify the nature of the patient’s deficits and serve as a tool for nonpharmacologic treatment planning, utilizing the patient’s cognitive strengths while minimizing the weaknesses.
The CLINICIAN’ S INITIAL IMPRESSION AND PLAN
Mr A had a 14-year history of Parkinson’s disease and had only recently developed cognitive symptoms as well as mild, intermittent visual hallucinations and illusions. He also had emotional lability. On the basis of the clinical presentation and the clinical history, the treating Banner Alzheimer’s Institute physician believed that Mr A was currently suffering from Parkinson’s disease dementia. Cognitive impairment in Parkinson’s disease tends to appear later in the course of the illness, as opposed to Lewy body dementia, in which cognitive impairment as well as visuospatial difficulties, hallucinations, and behavioral disturbances tend to appear fairly early in the course of the disease, typically within the first year after initial presentation of parkinsonian symptoms. However, further workup was necessary to rule out any intracranial pathology related to the patient’s cardiovascular risk factors, as well as a laboratory workup to rule out any metabolic factors contributing to his cognitive decline.
PLAN OF CARE
1. Due to the presence of metal in the body, a computed tomography (CT) scan of the head was ordered.2. Laboratory studies were requested from the patient’s primary care physician.3. Mr A was advised to continue treatment for Parkinson’s disease with his current neurologist. The potential side effects of antiparkinsonian therapy were discussed with Mr A and his wife, including the possibility of increased hallucinations and behavioral disturbances. They were advised to pay close attention to any increase in behavioral or psychotic symptoms following adjustments of parkinsonian medications or any physical or psychological stress.4. No medication adjustments were made at the time of the visit.5. Continue venlafaxine extended release at a dose of 150 mg daily.6. Continue clonazepam 0.5 mg nightly for insomnia. The potential disinhibitory effects of benzodiazepines in patients suffering from cognitive impairment were discussed with Mr A and his spouse. However, the physician felt that a full workup should be completed prior to further medication adjustment.7. No recommendations regarding hypersexuality were made at the time of the initial visit. Mr A was referred to the Family and Community Services team at Banner Alzheimer’s Institute for nonpharmacologic intervention regarding psychosocial and behavioral issues.
FOLLOW-UP APPOINTMENT
Mr A returned to the Stead Family Memory Clinic 1 month later. Although he continued to report a subjective cognitive decline, there had been no significant changes in functional abilities. Mobility was slightly worse with more gait instability. Increased irritability and emotional lability were also reported by Mr A’s wife.His wife reported that, during one episode, Mr A became very distraught over his current physical and cognitive impairments. He was physically and verbally aggressive toward her after she rejected his sexual advances stating that she no longer wished to engage in sexual activity. Mr A obtained a rifle from a storage area, loaded it, and pointed it at his wife. He then walked into the shower to kill himself. He was talked out of firing the gun by his wife, and he hid the gun in the garage. His wife stated that she did not call the police or 911 and had yet to discuss this event with a physician. He commented that he was “not even man enough” to kill himself. At the time of the follow-up visit, Mr A denied current active suicidal ideations but did admit to passive suicidal ideation, such as a wish to fall asleep and never wake up. He denied a current plan or intent to commit suicide.As per family report, Mr A continued to experience visual hallucinations of bugs; however, these were not disturbing to him. He also reported seeing family members in the home when they were not there. Mr A denied any paranoid ideations, homicidal ideations, auditory hallucinations, or command-type hallucinations.The head CT scan was unremarkable, only demonstrating mild periventricular white matter hypodensities. All laboratory studies were unremarkable.
What would you do next?
Your Colleagues Who Attended the Banner Alzheimer’s Institute Case Conference Answered as Follows:
| A. Watch and wait | 0% |
| B. Call the police and report the aborted suicide attempt | 0% |
| C. Increase the antidepressant | 0% |
| D. Start a different antidepressant | 0% |
| E. Admit the patient to an inpatient psychiatric unit | 80% |
| F. Start the NMDA antagonist (memantine) | 0% |
| G. Start an antipsychotic | 20% |
| H. Stop the carbidopa/levodopa | 0% |
The majority of those attending the case conference felt that inpatient psychiatric hospitalization would allow for the most rapid and aggressive treatment of the patient’s depressive symptoms as well as minimize the potential risk of additional suicide attempts. However, 20% of participants believed that aggressive outpatient antipsychotic treatment was a better option since Mr A was not currently an imminent danger to himself or others.
How would you address Mr A’s hypersexuality?
Your Colleagues Who Attended the Banner Alzheimer’s Institute Case Conference Answered as Follows:
| A. Couple’s counseling | 0% |
| B. Start an antidepressant | 40% |
| C. Start medroxyprogesterone acetate | 10% |
| D. Encourage regularly scheduled sexual activity and maintenance of a calendar | 0% |
| E. Provide gentleman’s magazine and encourage privacy for masturbation | 0% |
| F. Engage the patient in an adult day program | 0% |
| G. A and B | 0% |
| H. B and C | 30% |
| I. B, D, and E | 20% |
PLAN UPDATE
The physician at Banner Alzheimer’s Institute decided that Mr A was not currently an imminent threat to himself or others. Mr A was closely followed at the Stead Family Memory Clinic. He was seen by both the physician and the Family and Community Services team several times per month over the next several months. Pharmacologic changes were recommended in addition to behavioral and environmental modifications, the latter of which appeared to be most effective.1. Several different antidepressants (citalopram, desvenlafaxine, mirtazapine) were tried due to their potential benefit on emotional lability and depressive symptoms as well as their ability to reduce libido. Mr A and his wife saw limited effect with these medications.2. Mr A was started on memantine to slow the progression of cognitive and functional decline and to ameliorate his current emotional lability and increased impulsivity. The off-label use of this medication was discussed with the patient and his family. Memantine is currently approved for the moderate-to-severe stages of Alzheimer’s dementia; however, recent studies indicate that memantine may be of benefit for cognitive function and behavior in patients with Parkinson’s dementia (Emre et al, 2010).3. Due to the limited effect of antidepressant therapy and memantine and continued visual hallucinations, Mr A was started on quetiapine 25 mg bid. His spouse reported a moderate improvement in affective lability and impulsivity. No significant changes in visual hallucinations were reported. Attempts to increase the antipsychotic dose resulted in greater impairments in mobility. Individuals with parkinsonian syndromes tend to be more prone to the extrapyramidal side effects of antipsychotics. Hence, these medications need to be used with caution.The following nonpharmacologic interventions were recommended:1. Mr A’s wife was instructed to remove the gun and the ammunition from the home without the patient’s knowledge. If the family felt that they were unable to accomplish this task, they were instructed to call the police, which should not be done in front of Mr A.2. Mr A was referred to the Lee Silverman Voice Treatment “Big and Loud” classes (LSVT Global, 2013) to help improve mobility, gait, balance, and speech. As a result of the therapy, he was able to perform more tasks (dressing, personal care, feeding) independently, and his mood significantly improved.Physical, speech, and occupational programs are available for individuals suffering from parkinsonian syndromes. These programs, like Lee Silverman Voice Treatment, focus on “speaking loud” and “moving big.” The increased amplitude of limb and body movement (bigness) in people with parkinsonian syndromes like Parkinson’s dementia has shown documented improvements in amplitude (trunk rotation/gait) that translate to improved speed in the upper and lower limbs, balance, and quality of life. Speech therapy stimulates the motor speech system and incorporates sensory awareness training to help individuals with Parkinson’s disease recognize that their voice is too soft, convincing them that the louder voice is within normal limits and making them comfortable with their new louder voice.3. It was suggested that Mr A’s wife purchase a magazine subscription to a gentlemen’s magazine, which would allow the patient freedom and privacy to masturbate in his own room. This might be accompanied by other sexual tools. It was also suggested that a full-body pillow be placed in his bed on the side that his wife used to occupy.4. Mr A’s wife was encouraged not to rule out sexual activity entirely, but rather set a schedule that is marked on the calendar for 1-2 days per week. This method of tracking and planning sexual activity would enable Mr A to have more control and predictability, which could result in less perseveration on this topic.
DISCUSSION
Treatment of sexually challenging behaviors in patients with dementia requires a thorough understanding of behaviors in the context in which they occur, their frequency, and when and where they occur and with whom. An assessment of competency to engage in a sexual relationship may also be helpful for a clinician in deciding how to respond to sexual behaviors within an appropriate legal and ethical framework (Lichtenberg and Strzepek, 1990).Despite the fact that hypersexuality affects up to a fifth of all patients with dementia, there are currently no US Food and Drug-approved treatments or official guidelines for treating sexually disinhibited behaviors in dementia. The first step in treating hypersexuality in dementia is to thoroughly assess target behaviors and use environmental modifications and interventions.Nonpharmacologic interventions require a thorough assessment of problem behaviors, the context in which they occur, evaluation of who is distressed by the behavior, frequency, contributing factors, and associated risks. The decision-making capacity of the demented individual should also be thoroughly assessed.Although neurodegeneration of the frontal lobe systems is a biological factor affecting impulse control, psychological factors such as lack of physical closeness and loss of self-esteem and self-image should not be ignored. These factors can be addressed through nonsexual means such as alterations in communication patterns between the patient and caregiver. Use of comforting touch and other forms of emotional/psychic intimacy may also help alleviate the focus on physical sexual demands.Since there is very little published literature on pharmacologic treatment of hypersexuality in dementia, clinicians extrapolate from treatment studies of patients with paraphilias and sexual offenders (Grossman et al, 1999). However, none of these classes of agents have been shown to confer clinical benefit in people with dementia, and, in many cases, harm without benefit has been demonstrated in people with dementia (Burke and Tariot, 2010). Clinicians should only consider attempting empirical trials with caution and with a clearly delineated risk/benefit analysis. Currently, data on which to base pharmacologic treatment are insufficient to dictate best clinical practice; however, classes of drugs sometimes used include the following:
- Antipsychotics
- Serotonergics (citalopram, fluoxetine, clomipramine)
- Anticonvulsants
- Antiandrogens (medroxyprogesterone, cyproterone, cimetidine)
- Estrogens (estradiol, estrone, diethylstilbestrol)
- Luteinizing hormone-releasing hormone analogs (leuprolide, triptorelin, goserelin)
Individuals suffering from cognitive impairment frequently struggle with a sense of loss of control and self-esteem. Sexual performance historically enhances self-esteem. Sexual performance may also be a means of demonstrating control and mastery of a situation. Focusing on other means of enhancing self-esteem as functional impairment progresses over the course of the dementia may help reduce hypersexuality. In Mr A’s case, physical therapy was used to allow for more independence with day-to-day activities, which alleviated some of his subjective loss of autonomy.Since short-term memory problems are a key symptom in most dementias, individuals may forget immediate past sexual activity, resulting in repeated requests and advances toward sexual intercourse. Therefore, use of calendars to record when sexual activity occurred will serve as a physical reminder of sexual intercourse and allow patients a sense of predictability and control in their relationship.
Disclosure of off-label usage
The authors have determined that, to the best of their knowledge, memantine and donepezil are not approved by the US Food and Drug Administration for the treatment of Parkinson’s dementia, and estradiol, fluoxetine, goserelin, medroxyprogesterone acetate, mirtazapine, quetiapine, sertraline, triptorelin, cyproterone, diethylstilbestrol, estrone, and leuprolide are not approved for the treatment of hypersexuality in dementia.
FINANCIAL DISCLOSURE
Dr Yaari is a consultant for Amedisys Home Health. Dr Tariot has served as a consultant for Abbott, AC Immune, Adamas, Avanir, Boehringer-Ingelheim, Chase, Chiesi, Eisai, Elan, MedAvante, Merz, Neuroptix, Otsuka, and Sanofi-Aventis; has received consulting fees and research support from AstraZeneca, Avid, Bristol-Myers Squibb, Eli Lilly, Genentech, GlaxoSmithKline, Janssen, Medivation, Merck, Pfizer, Roche, and Toyama; has received research support only from Baxter, Functional Neuromodulation, GE, and Targacept; has received other research support from Alzheimer’s Association, Arizona Department of Health Services, National Institute of Mental Health, and National Institute on Aging; is a stock shareholder in Adamas; and is listed as a contributor to a patent owned by the University of Rochester (Rochester, New York) for “Biomarkers of Alzheimer’s Disease.” Drs Burke, Fleisher, and Hall and Ms Brand have no personal affiliations or financial relationships with any commercial interest to disclose relative to the activity.
FUNDING/SUPPORT
None reported.
CASE CONFERENCE
The Banner Alzheimer’s Institute Case Conference is a weekly event in which physicians and staff discuss challenging and/or teaching cases of patients seen at the Institute’s Stead Family Memory Clinic. These conferences are attended by a multidisciplinary group that includes Banner Alzheimer’s Institute dementia specialists, community physicians (internal medicine, family medicine, and radiology), physician assistants, social workers, nurses, medical students, residents, and fellows.
DISCLAIMER
The opinions expressed are those of the authors, not of Banner Health or Physicians Postgraduate Press, Inc.
Clinical Points
- Despite the fact that hypersexuality affects up to 17% of all patients with dementia, there are currently no US Food and Drug Administration-approved treatments or official guidelines for treating sexually disinhibited behaviors in dementia.
- Nonpharmacologic interventions should be attempted first and require a thorough assessment of problem behaviors, the context in which they occur, who is distressed by the behavior, frequency, contributing factors, and associated risks.
- Since little is published on pharmacologic treatment of hypersexuality in dementia, data are insufficient to dictate best clinical practice. Results extrapolated from treatment studies of patients with paraphilias and of sexual offenders may be helpful in guiding pharmacologic treatment.
This CME activity is expired. For more CME activities, visit cme.psychiatrist.com.
Find more articles on this and other psychiatry and CNS topics:
The Journal of Clinical Psychiatry
The Primary Care Companion for CNS Disorders
REFERENCES
Burke A, Tariot PN. Drug treatments for the behavioral and psychiatric symptoms of dementia. In: Ames D, Burns A, O’ Brien J, eds. Dementia. London, England: Hodder Arnold; 2010:231-252.Burns A, Jacoby R, Levy R. Psychiatric phenomena in Alzheimer’s disease, IV: disorders of behaviour. Br J Psychiatry. 1990;157(1):86-94. PubMeddoi:10.1192/bjp.157.1.86
Emre M, Tsolaki M, Bonuccelli U, et al; 11018 Study Investigators. Memantine for patients with Parkinson’s disease dementia or dementia with Lewy bodies: a randomized, double-blind, placebo-controlled trial. Lancet Neurol. 2010;9(10):969-977. PubMeddoi:10.1016/S1474-4422(10)70194-0
Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”: a practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12(3):189-198. PubMeddoi:10.1016/0022-3956(75)90026-6
Grossman LS, Martis B, Fichtner CG. Are sex offenders treatable? a research overview. Psychiatr Serv. 1999;50(3):349-361. PubMed
Johns EK, Phillips NA, Chertkow H, et al. The Montreal Cognitive Assessment: normative data in the community. In: Final Program of the 36th Annual Meeting of the International Neuropsychological Society; February 6-9, 2008; Waikoloa, Hawaii. J Int Neuropsychol Soc. 2008a;41(suppl 1):58.
Johns EK, Phillips NA, Chertkow H, et al. The effect of education on performance on the Montreal Cognitive Assessment (MoCA): normative data from the community [poster]. Canadian J Geriatrics. 2008b:11(1):62. Presented at the 28th Annual Meeting of the Canadian Geriatrics Society; April 2008; Montreal, Quebec, Canada
Lichtenberg PA, Strzepek DM. Assessments of institutionalized dementia patients’ competencies to participate in intimate relationships. Gerontologist. 1990;30(1):117-120. PubMeddoi:10.1093/geront/30.1.117
LSVT Global. Rehabilitative Therapy for People With Parkinson’s. http://www.lsvtglobal.com/index.php?action=home. Accessed October 15, 2013.Mungas D. In-office mental status testing: a practical guide. Geriatrics. 1991;46(7):54-58, 63, 66. PubMed
Nasreddine ZS, Phillips NA, Bédirian V, et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005;53(4):695-699. PubMeddoi:10.1111/j.1532-5415.2005.53221.xSeries D, Degano H. Hypersexuality in dementia. Adv Psychiatr Treat. 2005;11(6):424-431. doi:10.1192/apt.11.6.424
Please sign in or purchase this PDF for $40.00.
Save
Cite
