Objective: To determine whether specific depression symptoms are associated with glycemic control independent of potential demographic and clinical covariates among primary care patients with comorbid type 2 diabetes and provisional threshold or subthreshold depression.
Method: We examined a convenience sample of patients diagnosed with type 2 diabetes and provisional threshold or subthreshold depression (N = 82) at 2 family health centers. Cases were identified using a population-based registry of patients diagnosed with type 2 diabetes (ICD-9 codes 250.00 for controlled type 2 diabetes and 250.02 for uncontrolled type 2 diabetes). Data from patients with a primary care provider appointment from the beginning of April 2011 through the end of June 2012 and with at least one 9-item Patient Health Questionnaire (PHQ-9) depression screener and a glycated hemoglobin A1c (HbA1c) laboratory test between 2 weeks before and 10 weeks after PHQ-9 screening were eligible for inclusion. We defined provisional threshold or subthreshold depression using PHQ-9 scoring criteria, which were designed to yield provisional diagnostic information about major depressive disorder based on DSM-5 diagnostic criteria.
Results: Patients reporting higher severity of sleep problems on the PHQ-9 had significantly higher HbA1c levels (mean = 8.48, SD = 2.17) compared to patients reporting lower severity or absence of this symptom (mean = 7.19, SD = 1.34, t48.88 = −3.13, P = .003). Problems with sleep contributed unique variance on glycemic control (β = 0.27, P = .02) when controlling for potential clinical and demographic covariates, with those reporting more sleep difficulties having higher HbA1c levels.
Conclusions: For patients with type 2 diabetes and provisional threshold or subthreshold depression, it may be prudent to aggressively address sleep problems as a potential mechanism toward improving diabetes control.
Find more articles on this and other psychiatry and CNS topics:
The Journal of Clinical Psychiatry
The Primary Care Companion for CNS Disorders
CME Background
Articles are selected for credit designation based on an assessment of the educational needs of CME participants, with the purpose of providing readers with a curriculum of CME articles on a variety of topics throughout each volume. Activities are planned using a process that links identified needs with desired results.
To obtain credit, read the article, correctly answer the questions in the Posttest, and complete the Evaluation. The Posttest and Evaluation are available at http://www.cmeinstitute.com
/activities/Pages/journal.aspx.
CME Objective
After studying this article, you should be able to:
‘ ¢ Consider whether sleep problems contribute toward poor control of type 2 diabetes in patients with provisional threshold or subthreshold depression
Accreditation Statement
The CME Institute of Physicians Postgraduate Press, Inc., is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians.
Credit Designation
The CME Institute of Physicians Postgraduate Press, Inc., designates this journal-based CME activity for a maximum of 1.0 AMA PRA Category 1 Creditâ„¢. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
Note: The American Academy of Physician Assistants (AAPA) accepts certificates of participation for educational activities certified for AMA PRA Category 1 Creditâ„¢ from organizations accredited by ACCME or a recognized state medical society. Physician assistants may receive a maximum of 1.0 hour of Category I credit for completing this program.
Date of Original Release/Review
This educational activity is eligible for AMA PRA Category 1 Creditâ„¢ through October 31, 2017. The latest review of this material was September 2015.
Financial Disclosure
All individuals in a position to influence the content of this activity were asked to complete a statement regarding all relevant personal financial relationships between themselves or their spouse/partner and any commercial interest. The CME Institute has resolved any conflicts of interest that were identified. In the past year, Larry Culpepper, MD, MPH, Editor in Chief, has been a member of the speakers/advisory boards for AstraZeneca, Forest, Jazz, Lundbeck, Merck, Shire, Sunovion, and Takeda; has been a stock shareholder of M3 My Mood Monitor; and has received royalties from UpToDate and other financial or material support from Oxford University Press. No member of the CME Institute staff reported any relevant personal financial relationships. Faculty financial disclosure appears with the article.
Association Between Specific Depression Symptoms and Glycemic Control Among Patients With Comorbid Type 2 Diabetes and Provisional Depression
ABSTRACT
Objective: To determine whether specific depression symptoms are associated with glycemic control independent of potential demographic and clinical covariates among primary care patients with comorbid type 2 diabetes and provisional threshold or subthreshold depression.
Method: We examined a convenience sample of patients diagnosed with type 2 diabetes and provisional threshold or subthreshold depression (N = 82) at 2 family health centers. Cases were identified using a population-based registry of patients diagnosed with type 2 diabetes (ICD-9 codes 250.00 for controlled type 2 diabetes and 250.02 for uncontrolled type 2 diabetes). Data from patients with a primary care provider appointment from the beginning of April 2011 through the end of June 2012 and with at least one 9-item Patient Health Questionnaire (PHQ-9) depression screener and a glycated hemoglobin A1c (HbA1c) laboratory test between 2 weeks before and 10 weeks after PHQ-9 screening were eligible for inclusion. We defined provisional threshold or subthreshold depression using PHQ-9 scoring criteria, which were designed to yield provisional diagnostic information about major depressive disorder based on DSM-5 diagnostic criteria.
Results: Patients reporting higher severity of sleep problems on the PHQ-9 had significantly higher HbA1c levels (mean = 8.48, SD = 2.17) compared to patients reporting lower severity or absence of this symptom (mean = 7.19, SD = 1.34, t48.88 = −3.13, P = .003). Problems with sleep contributed unique variance on glycemic control (β = 0.27, P = .02) when controlling for potential clinical and demographic covariates, with those reporting more sleep difficulties having higher HbA1c levels.
Conclusions: For patients with type 2 diabetes and provisional threshold or subthreshold depression, it may be prudent to aggressively address sleep problems as a potential mechanism toward improving diabetes control.
Prim Care Companion CNS Disord 2015;17(5):doi:10.4088/PCC.14m01754
© Copyright 2015 Physicians Postgraduate Press, Inc.
aDepartment of Psychiatry, Rhode Island Hospital, Providence
bDepartment of Psychology, Suffolk University, Boston, Massachusetts
cPrimary Care, Providence VA Medical Center and Alpert Medical School, Brown University, Providence, Rhode Island
dDepartment of Quantitative Health Sciences, University of Massachusetts Medical School, Worcester
eDepartment of Family Medicine and Community Health, University of Massachusetts Medical School, Worcester
*Corresponding author: Stephanie J. Czech, PhD, Department of Psychiatry, Rhode Island Hospital, 146 West River St, Ste 11B, Providence, RI 02904 ([email protected]).
The prevalence rates of both type 2 diabetes and depression, disorders associated with debilitating functional impairment, have notably increased in recent decades.1,2 Moreover, both disorders commonly co-occur, as it is estimated that clinically relevant depression is nearly 2 times more likely in patients with type 2 diabetes than in those without.3 The comorbidity of the 2 illnesses profoundly impacts disease self-management, daily functioning, and quality of life4,5 and has been linked with diabetic complications, sexual dysfunction, and increased mortality,6 in addition to increased medical costs across medical and mental health settings.4
Comorbid depression may negatively impact the course and severity of diabetes via its influence on glycemic control.6-11 While mechanisms have not yet been determined,12,13 psychological/behavioral, sociocultural, and biological pathways have been proposed. For example, depression has been posited to interfere with good glycemic control via behavioral mechanisms, including inability to adhere to diabetes self-management recommendations.4,8 Biologically, research suggests that depression affects glycemic control via metabolically relevant pathways, including alterations in neuroendocrine or immune functioning and glucose metabolism.9,11 Likewise, poor glycemic control has been proposed to adversely affect mood, reinforcing this relationship.8 For example, high blood glucose levels in patients with diabetes have been found to be associated with negative mood states such as anger and sadness,14 increased worry about glucose status,15 and poor glycemic control with impaired sense of well-being and perceived health.16 On the biological side, there is some evidence to suggest that uncontrolled glycemia may impact mood through the effects of blood glucose fluctuations on cortical function in the brain.8,14
Several studies have examined the relationship between depression and glycemic control, and both cross-sectional and longitudinal studies have yielded inconsistent findings.10,13 While methodological issues may account for mixed findings, studies that include large populations in primary care and allow for the inclusion of confounding variables and potentially important moderators,4,17-19 have continued to exhibit inconsistent results.
To date, research examining the relationship between depression and glycemic control has treated depression as a homogenous construct. Yet, there is considerable evidence that depression is heterogeneous.20 For example, the DSM-521 criteria for depression allow for differences in not only the severity of symptoms (ie, mild, moderate, severe), but also the nature of symptoms (eg, negative emotions, anhedonia). Importantly, while heterogeneity of symptom patterns is not specific to depression, depression is unique in that somatic symptom criteria related to weight/appetite, sleep, and motor activity are bidirectional. In other words, individuals can meet these criteria for a depressive episode with either an increase or a decrease in these symptoms. Thus, examining relationships between depression symptoms and glycemic control may provide insight into specific aspects of depression most deleterious toward glycemic control in patients with type 2 diabetes.

- For patients with type 2 diabetes and depression, sleep problems may negatively impact diabetes control.
- There may be clinical benefit of deliberate assessment and intervention for sleep problems early in the treatment process for patients with type 2 diabetes and depression.
We have identified only 1 previous study examining the relationship between the 9 individual depression symptoms consistent with the DSM-5 diagnostic criteria and glycemic control. Bot and colleagues22 examined cross-sectional and longitudinal associations between 9-item Patient Health Questionnaire (PHQ-9) depressive symptoms and glycated hemoglobin A1c (HbA1c) in 343 and 291 patients with type 2 diabetes, respectively, finding a significant association with HbA1c for sleep problems, appetite problems, and suicidal ideation when adjusting for relevant controls at baseline and no association at 1-year follow-up. In that study,22 the sample was not limited to patients with probable clinically relevant depression, but instead included all diabetic patients who completed a PHQ-9 screener. In contrast, we utilized the PHQ-9 diagnostic algorithm, based on DSM-5 diagnostic criteria, to limit the sample to patients with provisional threshold and subthreshold depression. We believe this methodology provided an opportunity to detect significant predictors of HbA1c in a more clinically relevant sample of type 2 diabetes patients endorsing depression symptoms.
We aimed to determine whether specific depression symptoms are associated with glycemic control in primary care patients with comorbid type 2 diabetes and provisional threshold or subthreshold depression. We propose that identifying the most relevant depression symptoms in this population will serve to guide and focus interventions toward improving both depression and type 2 diabetes.
METHOD
Participants
Participants were a convenience sample of patients at 2 family health centers of the University of Massachusetts Memorial Medical Center in Worcester, Massachusetts, an industrial city, and Barre, Massachusetts, a rural town, both located in Central Massachusetts. The health centers serve as academic group practice model residency sites, emphasizing patient-centered full-spectrum family medicine care and family medicine residency education. The centers serve an ethnically and socioeconomically diverse population, consistent with the diversity of Worcester and the surrounding communities.
We identified cases using an established hospital-based registry of patients with a diagnosis of type 2 diabetes (ICD-9 codes 250.00 for controlled type 2 diabetes and 250.02 for uncontrolled type 2 diabetes). We included data from all patients who attended an appointment with their primary care provider from the beginning of April 2011 through the end of June 2012 and had a record of at least 1 PHQ-9 screen and an HbA1c laboratory test that occurred between 2 weeks before and 10 weeks after PHQ-9 screening. This time gap was intended to maximize the opportunity for temporal overlap between these measurements, given that HbA1c is a clinically useful index of mean blood glucose during the preceding 60 to 120 days23 and the PHQ-9 measures depression symptoms within the previous 2 weeks.24 Informed consent was waived by the University of Massachusetts Medical School Institutional Review Board according to the code of federal regulations that govern the protection of human subjects (45 CFR 46. 116 [d]) given that research was minimal risk, the waiver did not adversely affect the subject, and the research could not have practically been carried out without the waiver.
Procedure
As part of routine care, all patients with a diagnosis of type 2 diabetes were advised to have HbA1c tests and depression screening every 6 months. Chart alerts signaled the delivery of depression screeners to all patients with a diabetes diagnosis at the time of appointment check in. Medical assistants were trained to assist patients in screener completion if necessary, and screeners were collected by primary care physicians during patient visits. Screener data were subsequently scanned into the electronic health record, and individual item responses were entered into a database that supported patient care for patients with type 2 diabetes and other chronic health problems, including major depressive disorder.
Demographic, health behavior, and clinical data were entered into the electronic health record through the registration process via the registration department within the University of Massachusetts Memorial Medical Center system (ie, race, ethnicity, age, gender, employment status, marital status), by primary care physicians during the medical visit (ie, diabetes complications, medical comorbidity, medication regimen, body mass index [BMI], smoking status), and by the office staff at time of check in for the medical visit (ie, health insurance information).
Measures
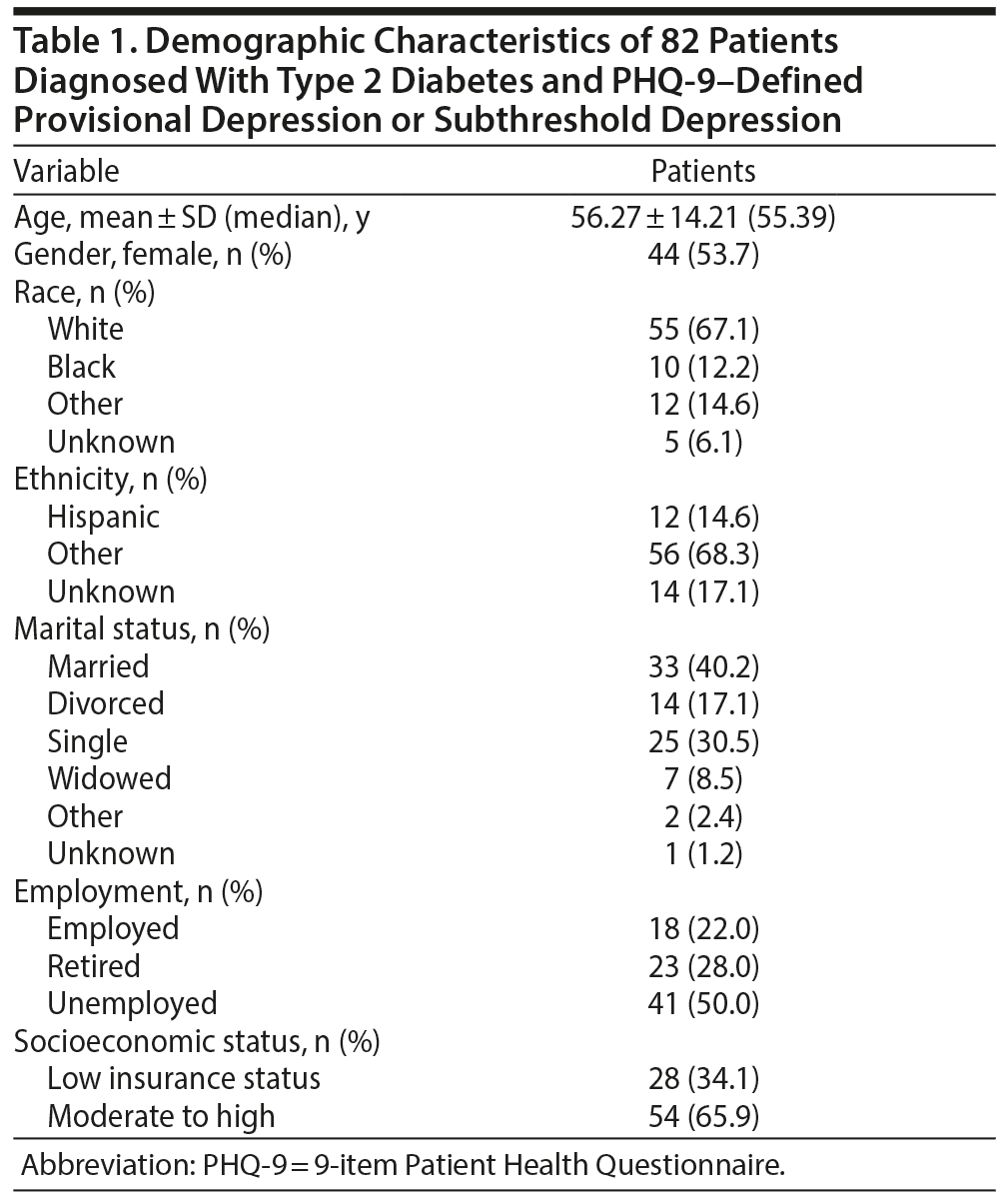
Demographic characteristics. Information on demographic characteristics was collected by patient self-report and included race (ie, white, African American/black, other, unknown), ethnicity (ie, Hispanic, other, unknown), age (in years), gender (ie, male, female), socioeconomic status (as defined by health insurance information, ie, “low insurance status” = Mass Health, Commonwealth Care, Health Safety Net; “moderate to high insurance status” = private health insurance, Commonwealth Choice, Medicare; unknown = unknown), employment status (ie, employed, student, unemployed, retired, unknown), and marital status (ie, married, divorced, single, widowed, other, unknown).
Provisional depression. We assessed depressive symptoms with the PHQ-9, a 9-item self-report depression module taken from the full PHQ,25 which covers 8 diagnostic categories and was developed to correspond to specific DSM-IV-R26 diagnoses. The PHQ-9 items are consistent with the recently revised DSM-5, and the American Psychiatric Association has recommended continued use of the measure to enhance clinical decision-making.21 The PHQ-9 is a dual-purpose instrument that yields provisional diagnostic information about threshold and subthreshold MDD diagnoses and can be used as a severity measure to grade depressive symptom severity and monitor patient status over time.24,25 The PHQ-9 has been found to have strong psychometric properties,24,25,27 and health care guidelines strongly support its use with patients diagnosed with chronic diseases including diabetes.28,29 The Cronbach α coefficient on the PHQ-9 for the current study was found to be 0.75.
Items on the PHQ-9 reflect the 9 diagnostic criteria of depression and are scored from 0 (“not at all”) to 3 (“nearly every day”). Scores range from 0 to 27, with higher scores reflecting greater depression severity. Cutpoints of 5, 10, 15, and 20 represent thresholds for mild, moderate, moderately severe, and severe depression symptoms, respectively. When used as a diagnostic measure, 8 of the 9 symptoms count toward a provisional diagnosis of MDD if they are endorsed as present for at least “more than half the days” (score ≥ 2) in the past 2 weeks. The item assessing suicidal ideation counts toward a provisional diagnosis if it is present at all during the previous 2 weeks (score ≥ 1). A provisional diagnosis of MDD requires the presence of ≥ 5 symptoms (which must include either depressed mood or anhedonia). A provisional diagnosis of subthreshold depression requires ≥ 2 symptoms (including depressed mood or anhedonia) to be endorsed.
Health behavior risk factors. We collected BMI (kg/m2) measurements and patient self-report of status as a smoker from the medical chart.
Glycemic control. We obtained HbA1c levels and dates of each measurement from laboratory test results as reported in the medical chart.
Medical comorbidity. We measured medical comorbidity with the Charlson Comorbidity Index,30 a weighted index reflecting the extent of comorbidity considering 19 medical conditions, which has been used in previous research on the association between depression and glycemic control in primary care.19 This empirically derived measure was developed from a cohort of 604 patients admitted during a 1-month period to assess the risk of death from comorbid disease. Adjusted relative risks were used to derive weights (0 to 6) for 19 medical conditions. The total score for each patient is equal to the total sum of the weights of each condition listed as diagnoses (and identified by ICD-9 codes) in the electronic health record for that patient (ie, higher total score suggests a higher risk of death from comorbid disease). We employed an updated ICD-9-Clinical Modification coding algorithm consistent with Quan et al31 and excluded 2 conditions—diabetes and diabetes with end organ damage—from this measure (given that study inclusion criteria already required a diagnosis of diabetes, and a separate measure, as described below, captured the presence of diabetes complications such as end organ damage). Thus, for the present study, the item count was reduced to 17 items, with a possible total score of 32.
Severity of diabetes complications. We modeled diabetes complications with the Diabetes Complications Severity Index (DCSI),32 a scale used in previous research,18 which integrates data from diagnostic codes (ICD-9), pharmacy, and laboratory results to model the severity of diabetes complications from 7 categories (ie, retinopathy, nephropathy, neuropathy, cerebrovascular disease, cardiovascular disease, peripheral vascular disease, and metabolic syndrome) at any 1 point in time. Each category of complication is associated with a list of ICD-9 diagnoses, and each diagnosis was given a severity rating score based on previous models. Complication scores range from 0 to 80 and depend on the presence of specific ICD-9 codes in the electronic health record for a given patient.
Complexity of medication regimen. We extracted data concerning medications from the electronic health record to measure complexity of the medication regimen for diabetes (ie, diet only, oral hypoglycemic only, insulin only, or oral hypoglycemic plus insulin). The electronic health record medication list was reconciled by the primary care physician at every patient visit. If a patient was not prescribed any medications, then the classification “diet and exercise only” was assigned for this variable.
Analyses
We conducted 9 independent sample t tests to explore potential differences in HbA1c between patients indicating higher severity (ie, score of 2 or 3) versus absent or lower severity (ie, score of 0 or 1, respectively) of each of the original 9 PHQ-9 items. For each item that yielded a significant group difference, we employed Pearson product-moment correlations to examine the magnitude of the relationship between PHQ-9 items and HbA1c. Pearson product-moment correlations were also conducted between demographic and clinical variables and HbA1c. We conducted multiple regression to examine unique contributions of specific PHQ-9 items to HbA1c. In all analyses, model assumptions were reasonably upheld for all statistical tests, thus no data transformations were necessary.
RESULTS
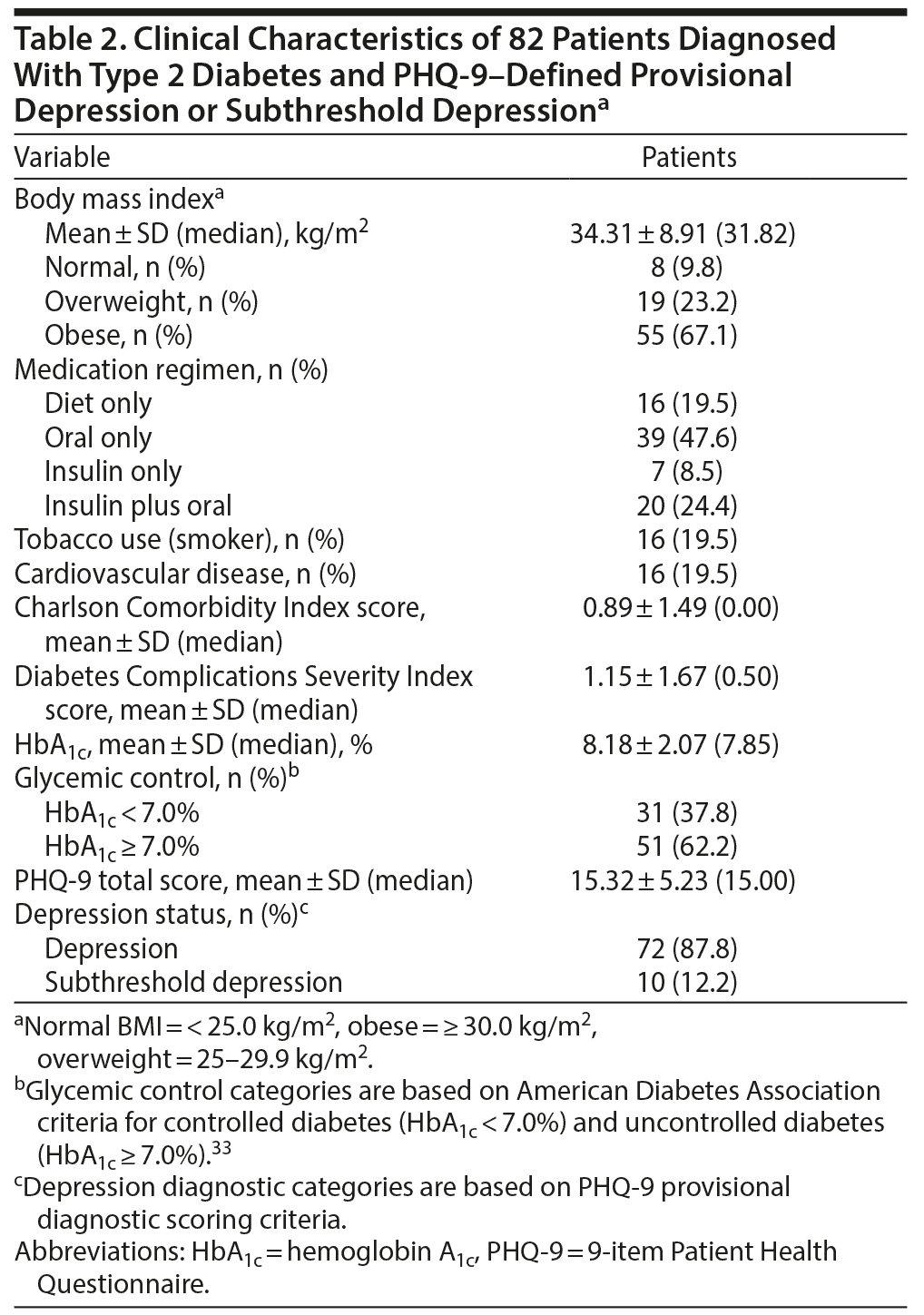
Data from 82 adults (44 females) meeting the inclusion criteria for the current study were analyzed. Demographic and clinical characteristics for the total sample are described in Tables 1 and 2.
We conducted 9 independent-sample t tests using the Bonferroni correction comparing HbA1c levels between groups of patients reporting high severity (ie, score of 2 or 3) versus low severity (ie, score of 0 or 1) of symptoms on each of the 9 PHQ-9 items. For PHQ-9 item 3 related to sleep problems (ie, “Trouble falling or staying asleep, or sleeping too much?”), patients in the high symptom severity group were found to have significantly higher HbA1c levels (mean = 8.48, SD = 2.17) compared to those in the low symptom severity group (mean = 7.19, SD = 1.34, t48.88 = −3.13, P = .003). The magnitude of the difference was large (η2 = 0.11). No other significant differences emerged.
To further explore the potential relationship between sleep problems and HbA1c, we conducted Pearson product moment correlation. A small but significant association was found between sleep problems and HbA1c (r = 0.23, n = 82, P = .04).
To examine the potentially unique contribution of sleep problems to HbA1c, we conducted multiple regression analysis (Table 3). Demographic and clinical variables were employed that have been identified in the literature as moderators of the relationship between depression and HbA1c as covariates (ie, comorbid cardiovascular disease, age, and gender). While other covariates were considered, we did not include them in the model given that they were not significantly correlated with HbA1c. The overall equation was significant (R2 = 0.14, F4,77 = 3.05, P = .02), and sleep problems contributed unique variance on HbA1c (β = 0.27, P = .02), with those reporting more sleep difficulties having higher HbA1c levels.
CONCLUSION
We sought to determine whether specific symptoms of depression are associated with glycemic control in a population of primary care patients with comorbid type 2 diabetes and provisional threshold or subthreshold depression. We found glycemic control varied with severity of PHQ-9 symptom only for the item related to sleep problems. Further exploration of this relationship revealed a unique contribution of sleep problems to HbA1c, with patients reporting more sleep difficulties having significantly higher HbA1C levels.
Strengths of our study include limiting the sample to patients with provisional threshold and subthreshold depression, allowing for a clinically relevant population. Additional strengths include data collection in the primary care setting wherein most patients with type 2 diabetes present, considering potentially confounding clinical and demographic variables for analyses, and specifying the timing of depression screening and HbA1c measurement in order to maximize the opportunity for temporal overlap between these measurements. Limitations include the cross-sectional nature of the study design, small sample size, inability to stratify our data based on meeting criteria for provisional threshold versus subthreshold depression due to inadequate power, utilization of a convenience sample of primarily white patients with moderate to high socioeconomic status, and lack of clinical interview, which would more accurately diagnose depression and its subtypes. Further, the false-positive rate for PHQ-9 measurement of threshold depression (sensitivity = 0.80, 95% CI, 0.71-0.87)27 may have obscured the presence of depression in our sample.
Research to date on the relationship between depression and glycemic control in patients with type 2 diabetes has been inconsistent.10,13 A possible explanation is that studies have not accounted for the heterogeneity of depression symptoms. In terms of individual symptoms of depression, our study suggests that, specifically, sleep problems are related to elevated HbA1c levels in type 2 diabetic patients with provisional subthreshold or threshold depression. We were unable to assess the bidirectional somatic symptoms of sleep, appetite, and motor activity in this study, and, thus, the question of whether specific sleep problems (ie, insomnia vs hypersomnia) may differentially relate to glycemic control in this population suggests a further line of investigation. Of note, 2 relatively common DSM-5 depression subtypes, atypical depression and melancholic depression, have been well validated in the literature and may be particularly relevant in patients with type 2 diabetes given their opposite patterns of somatic symptoms pertaining to sleep and appetite.34,35 Thus, future research might investigate the relationship between depression subtypes and HbA1c in patients with type 2 diabetes.
Our findings have clinical implications concerning depressed patients with type 2 diabetes presenting in the primary care clinic. Sleep problems are commonly reported in patients with diabetes, and studies have associated diabetes with poor sleep quality, sleep duration (due to either insomnia or sleep-disordered breathing), and increased daytime sleepiness.36-39 Further, there is accumulating evidence supporting associations between poor sleep quality and duration (including due to obstructive sleep apnea) and poor glycemic control in patients with type 2 diabetes when controlling for important potential confounds.40,41 Research examining the impact of improved sleep on glycemic control is sparse. While studies addressing the impact of continuous positive airway pressure therapy on glycemic control in diabetic patients with sleep apnea are accumulating, findings are mixed and long-term treatment studies are needed.42
With respect to depression, poor sleep is a well-known risk factor and maintaining factor.43 Ample empirical evidence supports the use of behavioral therapies and medications in treating insomnia in the context of depression.44 Randomized clinical trials45-47 further indicate that treating both insomnia and depression simultaneously leads to higher depression remission rates than treating depression alone.
Clinically, a question of interest is whether and how improving sleep may impact both depression severity and diabetes control in patients with this comorbidity. To answer this question, it will be important to understand mechanisms by which sleep disturbances potentially contribute to poor glycemic control in depressed patients. It appears that from a pathophysiologic standpoint, uncontrolled diabetes, depression, and insufficient sleep duration and quality may have commonalities (ie, concerning activation of the sympathetic nervous system, hypothalamic-pituitary-adrenal axis, and inflammatory processes).9,11,48 Thus, it is possible that these processes exacerbate and influence the effect of these comorbidities on each other. Importantly, sleep disorders (including short sleep duration and obstructive sleep apnea) and depression are both associated with obesity,49,50 which is a well-studied risk factor for impaired metabolic control with potential behavioral (eg, daytime fatigue and physical inactivity, increased appetite and eating behavior), psychological (eg, increase in emotional eating), and pathophysiologic mechanisms.48 Finally, behaviorally there are several ways in which sleep problems may negatively impact one’s ability to obtain optimal targets for glycemic control above and beyond the potentially disrupting effects of depressed mood and anhedonia. For example, severe fatigue and excess daytime sleep would most likely negatively affect one’s ability to effectively comply with diabetes self-management recommendations.
There has been recent attention toward tailoring interventions for depression in patients with diabetes. While cognitive-behavioral therapy, antidepressant medication, and collaborative care interventions have been found to be effective for improving depression in this population, findings are mixed in terms of their effectiveness toward improving glycemic control.51 Our findings suggest that for patients with type 2 diabetes and provisional depression or subthreshold depression, there may be clinical benefit toward deliberate assessment and intervention for sleep problems early in the treatment process. For patients in this population, sleep may be a mechanism toward improving diabetes control.
Submitted: May 22, 2014; accepted May 28, 2015.
Published online: October 15, 2015.
Disclosure of off-label usage: The authors have determined that, to the best of their knowledge, no investigational information about pharmaceutical agents that is outside US Food and Drug Administration-approved labeling has been presented in this article.
Financial disclosure: Drs Czech, Orsillo, Pirraglia, English, and Connell have no personal affiliations or financial relationships with any commercial interest to disclose relevant to the article.
Funding/support: None reported.
Acknowledgments: The authors thank Alexander Blount, EdD; Christine Runyan, PhD; and Daniel Mullin, PsyD (Department of Family Medicine and Community Health, University of Massachusetts Medical School, Worcester) for their support of this project at Hahnemann Family Health Center and Barre Family Health Center and Sara Sullivan, MA (Sullivan Evaluation Services, Orlando, Florida), for her technical assistance concerning data merging. Drs Blount, Runyan, and Mullin and Ms Sullivan have no personal affiliations or financial relationships with any commercial interest to disclose relevant to the article.
Find more articles on this and other psychiatry and CNS topics:
The Journal of Clinical Psychiatry
The Primary Care Companion for CNS Disorders
REFERENCES
1. Geiss LS, Pan L, Cadwell B, et al. Changes in incidence of diabetes in US adults, 1997-2003. Am J Prev Med. 2006;30(5):371-377. PubMed doi:10.1016/j.amepre.2005.12.009
2. Kessler RC, Berglund P, Demler O, et al; National Comorbidity Survey Replication. The epidemiology of major depressive disorder: results from the National Comorbidity Survey Replication (NCS-R). JAMA. 2003;289(23):3095-3105. PubMed doi:10.1001/jama.289.23.3095
3. Ali S, Stone MA, Peters JL, et al. The prevalence of co-morbid depression in adults with Type 2 diabetes: a systematic review and meta-analysis. Diabet Med. 2006;23(11):1165-1173. PubMed doi:10.1111/j.1464-5491.2006.01943.x
4. Ciechanowski PS, Katon WJ, Russo JE. Depression and diabetes: impact of depressive symptoms on adherence, function, and costs. Arch Intern Med. 2000;160(21):3278-3285. PubMed doi:10.1001/archinte.160.21.3278
5. Jacobson AM, de Groot M, Samson JA. The effects of psychiatric disorders and symptoms on quality of life in patients with type I and type II diabetes mellitus. Qual Life Res. 1997;6(1):11-20. PubMed doi:10.1023/A:1026487509852
6. de Groot M, Anderson R, Freedland KE, et al. Association of depression and diabetes complications: a meta-analysis. Psychosom Med. 2001;63(4):619-630. PubMed doi:10.1097/00006842-200107000-00015
7. Lustman PJ, Clouse RE. Treatment of depression in diabetes: impact on mood and medical outcome. J Psychosom Res. 2002;53(4):917-924. PubMed doi:10.1016/S0022-3999(02)00416-6
8. Lustman PJ, Clouse RE. Depression in diabetic patients: the relationship between mood and glycemic control. J Diabetes Complications. 2005;19(2):113-122. PubMed
9. Musselman DL, Betan E, Larsen H, et al. Relationship of depression to diabetes types 1 and 2: epidemiology, biology, and treatment. Biol Psychiatry. 2003;54(3):317-329. PubMed doi:10.1016/S0006-3223(03)00569-9
10. Georgiades A, Zucker N, Friedman KE, et al. Changes in depressive symptoms and glycemic control in diabetes mellitus. Psychosom Med. 2007;69(3):235-241. PubMed doi:10.1097/PSY.0b013e318042588d
11. Rustad JK, Musselman DL, Nemeroff CB. The relationship of depression and diabetes: pathophysiological and treatment implications. Psychoneuroendocrinology. 2011;36(9):1276-1286. PubMed doi:10.1016/j.psyneuen.2011.03.005
12. Llorente MD, Malphurs JE. Diabetes and Depression. In: Biessels GJ, Luchsinger LG, eds. Diabetes and the Brain. New York, NY: Springer; 2009:343-363. doi:10.1007/978-1-60327-850-8_14
13. Lustman PJ, Anderson RJ, Freedland KE, et al. Depression and poor glycemic control: a meta-analytic review of the literature. Diabetes Care. 2000;23(7):934-942. PubMed doi:10.2337/diacare.23.7.934
14. Gonder-Frederick LA, Cox DJ, Bobbitt SA, et al. Mood changes associated with blood glucose fluctuations in insulin-dependent diabetes mellitus. Health Psychol. 1989;8(1):45-59. PubMed doi:10.1037/0278-6133.8.1.45
15. Savli H, Sevinc A. The evaluation of the Turkish version of the Well-being Questionnaire (WBQ-22) in patients with Type 2 diabetes: the effects of diabetic complications. J Endocrinol Invest. 2005;28(10):683-691. PubMed doi:10.1007/BF03347550
16. Van der Does FE, De Neeling JN, Snoek FJ, et al. Symptoms and well-being in relation to glycemic control in type II diabetes. Diabetes Care. 1996;19(3):204-210. PubMed doi:10.2337/diacare.19.3.204
17. Katon WJ, Lin EH, Russo J, et al. Cardiac risk factors in patients with diabetes mellitus and major depression. J Gen Intern Med. 2004;19(12):1192-1199. PubMed doi:10.1111/j.1525-1497.2004.30405.x
18. Katon W, von Korff M, Ciechanowski P, et al. Behavioral and clinical factors associated with depression among individuals with diabetes. Diabetes Care. 2004;27(4):914-920. PubMed doi:10.2337/diacare.27.4.914
19. Trief PM, Morin PC, Izquierdo R, et al. Depression and glycemic control in elderly ethnically diverse patients with diabetes: the IDEATel project. Diabetes Care. 2006;29(4):830-835. PubMed doi:10.2337/diacare.29.04.06.dc05-1769
20. Hasler G, Drevets WC, Manji HK, et al. Discovering endophenotypes for major depression. Neuropsychopharmacology. 2004;29(10):1765-1781. PubMed doi:10.1038/sj.npp.1300506
21. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition. Arlington, VA: American Psychiatric Publishing; 2013.
22. Bot M, Pouwer F, de Jonge P, et al. Differential associations between depressive symptoms and glycaemic control in outpatients with diabetes. Diabet Med. 2013;30(3):e115-e122. PubMed doi:10.1111/dme.12082
23. Sacks DB, Arnold M, Bakris GL, et al; Evidence-Based Laboratory Medicine Committee of the American Association for Clinical Chemistry. Guidelines and recommendations for laboratory analysis in the diagnosis and management of diabetes mellitus. Diabetes Care. 2011;34(6):e61-e99. PubMed doi:10.2337/dc11-9998
24. Kroenke K, Spitzer RL, Williams JBW. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16(9):606-613. PubMed doi:10.1046/j.1525-1497.2001.016009606.x
25. Spitzer RL, Kroenke K, Williams JBW; Patient Health Questionnaire. Validation and utility of a self-report version of PRIME-MD: the PHQ primary care study. Primary Care Evaluation of Mental Disorders. JAMA. 1999;282(18):1737-1744. PubMed doi:10.1001/jama.282.18.1737
26. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision. Washington, DC: American Psychiatric Association; 2000.
27. Gilbody S, Richards D, Brealey S, et al. Screening for depression in medical settings with the Patient Health Questionnaire (PHQ): a diagnostic meta-analysis. J Gen Intern Med. 2007;22(11):1596-1602. PubMed doi:10.1007/s11606-007-0333-y
28. American Psychiatric Association. Practice Guideline for the Treatment of Patients With Major Depressive Disorder. 3rd ed. Arlington, VA: American Psychiatric Association; 2010
29. Mitchell J, Trangle M, Degnan B, et al. Health care guideline: adult depression in primary care. Institute for Clinical Systems Improvement Web site. https://www.icsi.org/_asset/fnhdm3/Depr-Interactive0512b.pdf. Updated September 2013. Accessed July 9, 2015.
30. Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40(5):373-383. PubMed doi:10.1016/0021-9681(87)90171-8
31. Quan H, Sundararajan V, Halfon P, et al. Coding algorithms for defining comorbidities in ICD-9-CM and ICD-10 administrative data. Med Care. 2005;43(11):1130-1139. PubMed doi:10.1097/01.mlr.0000182534.19832.83
32. Young BA, Lin E, Von Korff M, et al. Diabetes complications severity index and risk of mortality, hospitalization, and healthcare utilization. Am J Manag Care. 2008;14(1):15-23. PubMed
33. American Diabetes Association. Standards of medical care in diabetes—2014. Diabetes Care. 2014;37(suppl 1):S14-S80. PubMed
34. Lamers F, de Jonge P, Nolen WA, et al. Identifying depressive subtypes in a large cohort study: results from the Netherlands Study of Depression and Anxiety (NESDA). J Clin Psychiatry. 2010;71(12):1582-1589. PubMed doi:10.4088/JCP.09m05398blu
35. Gold PW, Gabry KE, Yasuda MR, et al. Divergent endocrine abnormalities in melancholic and atypical depression: clinical and pathophysiologic implications. Endocrinol Metab Clin North Am. 2002;31(1):37-62, vi. PubMed doi:10.1016/S0889-8529(01)00022-6
36. Einhorn D, Stewart DA, Erman MK, et al. Prevalence of sleep apnea in a population of adults with type 2 diabetes mellitus. Endocr Pract. 2007;13(4):355-362. PubMed doi:10.4158/EP.13.4.355
37. Gislason T, Almqvist M. Somatic diseases and sleep complaints: an epidemiological study of 3,201 Swedish men. Acta Med Scand. 1987;221(5):475-481. PubMed doi:10.1111/j.0954-6820.1987.tb01283.x
38. Resnick HE, Redline S, Shahar E, et al; Sleep Heart Health Study. Diabetes and sleep disturbances: findings from the Sleep Heart Health Study. Diabetes Care. 2003;26(3):702-709. PubMed doi:10.2337/diacare.26.3.702
39. Sridhar GR, Madhu K. Prevalence of sleep disturbances in diabetes mellitus. Diabetes Res Clin Pract. 1994;23(3):183-186. PubMed doi:10.1016/0168-8227(94)90103-1
40. Aronsohn RS, Whitmore H, Van Cauter E, et al. Impact of untreated obstructive sleep apnea on glucose control in type 2 diabetes. Am J Respir Crit Care Med. 2010;181(5):507-513. PubMed doi:10.1164/rccm.200909-1423OC
41. Knutson KL, Ryden AM, Mander BA, et al. Role of sleep duration and quality in the risk and severity of type 2 diabetes mellitus. Arch Intern Med. 2006;166(16):1768-1774. PubMed doi:10.1001/archinte.166.16.1768
42. Punjabi NM; Workshop Participants. Do sleep disorders and associated treatments impact glucose metabolism? Drugs. 2009;69(suppl 2):13-27. PubMed doi:10.2165/11531150-000000000-00000
43. Manber R, Chambers AS. Insomnia and depression: a multifaceted interplay. Curr Psychiatry Rep. 2009;11(6):437-442. PubMed doi:10.1007/s11920-009-0066-1
44. Jindal RD, Thase ME. Treatment of insomnia associated with clinical depression. Sleep Med Rev. 2004;8(1):19-30. PubMed doi:10.1016/S1087-0792(03)00025-X
45. Fava M, McCall WV, Krystal A, et al. Eszopiclone co-administered with fluoxetine in patients with insomnia coexisting with major depressive disorder. Biol Psychiatry. 2006;59(11):1052-1060. PubMed doi:10.1016/j.biopsych.2006.01.016
46. Manber R, Edinger JD, Gress JL, et al. Cognitive behavioral therapy for insomnia enhances depression outcome in patients with comorbid major depressive disorder and insomnia. Sleep. 2008;31(4):489-495. PubMed
47. Watanabe N, Furukawa TA, Shimodera S, et al. Brief behavioral therapy for refractory insomnia in residual depression: an assessor-blind, randomized controlled trial. J Clin Psychiatry. 2011;72(12):1651-1658. PubMed doi:10.4088/JCP.10m06130gry
48. Barone MTU, Menna-Barreto L. Diabetes and sleep: a complex cause-and-effect relationship. Diabetes Res Clin Pract. 2011;91(2):129-137. PubMed doi:10.1016/j.diabres.2010.07.011
49. Luppino FS, de Wit LM, Bouvy PF, et al. Overweight, obesity, and depression: a systematic review and meta-analysis of longitudinal studies. Arch Gen Psychiatry. 2010;67(3):220-229. PubMed doi:10.1001/archgenpsychiatry.2010.2
50. Spiegel K, Tasali E, Leproult R, et al. Effects of poor and short sleep on glucose metabolism and obesity risk. Nat Rev Endocrinol. 2009;5(5):253-261. PubMed doi:10.1038/nrendo.2009.23
51. Markowitz SM, Gonzalez JS, Wilkinson JL, et al. A review of treating depression in diabetes: emerging findings. Psychosomatics. 2011;52(1):1-18. PubMed doi:10.1016/j.psym.2010.11.007