Objective: Obsessive-compulsive symptoms (OCS) may be underrecognized in patients suffering from major depressive disorder. These patients may not receive optimal psychopharmacologic or psychological treatment if their OCS are not attended to. We performed a secondary analysis of the Sequenced Treatment Alternatives to Relieve Depression (STAR*D) trial database, the largest effectiveness study of “real-world” depression ever conducted, to determine the frequency of OCS and their effects on depression outcome.
Method: 3,984 adult subjects without a previous selective serotonin reuptake inhibitor trial for their current major depressive episode, per DSM-IV diagnostic criteria, had data for rating scales of interest at entry into Level 1 of the STAR*D trial, a 12-week open trial of citalopram at a dose of 20-60 mg/d to assess rates of depression remission. Our primary interest was the OCD subscale of the Psychiatric Diagnostic Screening Questionnaire.
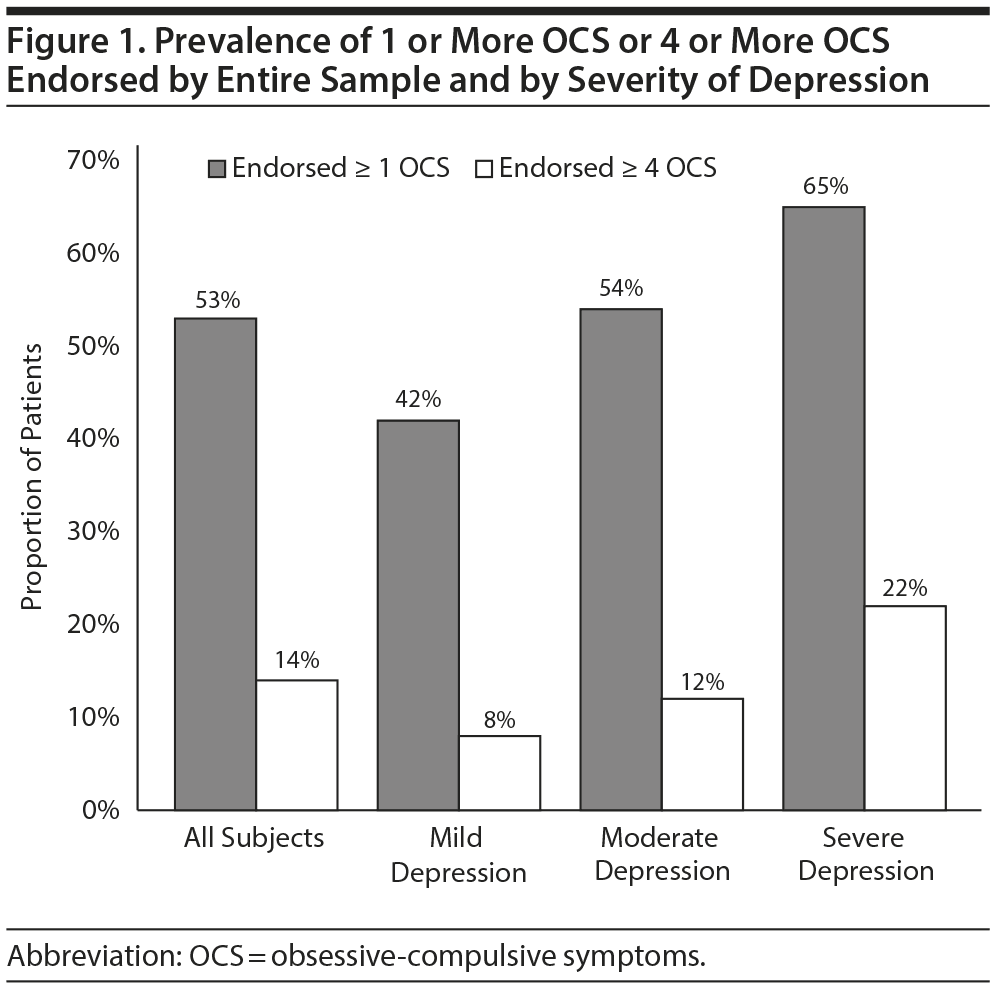
Results: At study entry, 53% of the STAR*D sample (which excluded patients with primary obsessive-compulsive disorder) endorsed ≥ 1 OCS, and 14% endorsed ≥ 4 OCS. Subjects endorsing ≥ 4 OCS had significantly lower corrected odds of depression remission on both the 17-item Hamilton Depression Rating Scale (HDRS-17) (OR = 0.61) and the Quick Inventory of Depressive Symptomatology (odds ratio [OR] = 0.51). Number of OCS endorsed was positively correlated with HDRS-17 score (r = 0.26, P < .0001). Consistent with findings from the Dunedin, New Zealand Community Study, the most common obsessions were doubts of having inadvertently caused harm and violent obsessions.
Conclusions: OCS are common in depressed outpatients and are often not attended to. They impact clinical recovery from depression and should be screened for since sufferers are often reluctant to disclose these symptoms.
Trial Registration: ClinicalTrials.gov identifier: NCT00021528

ABSTRACT
Objective: Obsessive-compulsive symptoms (OCS) may be underrecognized in patients suffering from major depressive disorder. These patients may not receive optimal psychopharmacologic or psychological treatment if their OCS are not attended to. We performed a secondary analysis of the Sequenced Treatment Alternatives to Relieve Depression (STAR*D) trial database, the largest effectiveness study of “real-world” depression ever conducted, to determine the frequency of OCS and their effects on depression outcome.
Method: 3,984 adult subjects without a previous selective serotonin reuptake inhibitor trial for their current major depressive episode, per DSM-IV diagnostic criteria, had data for rating scales of interest at entry into Level 1 of the STAR*D trial, a 12-week open trial of citalopram at a dose of 20-60 mg/d to assess rates of depression remission. Our primary interest was the OCD subscale of the Psychiatric Diagnostic Screening Questionnaire.
Results: At study entry, 53% of the STAR*D sample (which excluded patients with primary obsessive-compulsive disorder) endorsed ≥ 1 OCS, and 14% endorsed ≥ 4 OCS. Subjects endorsing ≥ 4 OCS had significantly lower corrected odds of depression remission on both the 17-item Hamilton Depression Rating Scale (HDRS-17) (OR = 0.61) and the Quick Inventory of Depressive Symptomatology (odds ratio [OR] = 0.51). Number of OCS endorsed was positively correlated with HDRS-17 score (r = 0.26, P < .0001). Consistent with findings from the Dunedin, New Zealand Community Study, the most common obsessions were doubts of having inadvertently caused harm and violent obsessions.
Conclusions: OCS are common in depressed outpatients and are often not attended to. They impact clinical recovery from depression and should be screened for since sufferers are often reluctant to disclose these symptoms.
Trial Registration: ClinicalTrials.gov identifier: NCT00021528
J Clin Psychiatry 2015;76(12):1668-1674
dx.doi.org/10.4088/JCP.14m09670
© Copyright 2015 Physicians Postgraduate Press, Inc.
aDepartment of Psychiatry, Massachusetts General Hospital, Boston
bDepartment of Psychiatry, University of Texas Southwestern Medical Center, Dallas
cDuke-National University of Singapore, Singapore
dHealth Policy Institute, University of Pittsburgh, Pittsburgh, Pennsylvania
*Corresponding author: Lee Baer, PhD, Massachusetts General Hospital, 1 Bowdoin Sq, 6th floor, Boston, MA 02114 ([email protected]).
Obsessive-compulsive disorder (OCD) imposes a severe burden of illness on its sufferers around the world.1-3 Yet, obsessive-compulsive symptoms (OCS) remain widely underrecognized or misdiagnosed by mental health professionals around the world: only 28% of patients meeting DSM-IV criteria for OCD at 14 German psychiatric clinics had been diagnosed with this problem,4 and when mental health professionals were presented with vignettes of patients with OCS, 77% misdiagnosed homosexual obsessions and 43% misdiagnosed aggressive obsessions.5 One reason for the underrecognition of OCS is that sufferers are particularly reluctant to discuss repugnant, embarrassing, and stigmatized violent or sexual obsessions with treaters: among Spanish OCD sufferers, the main barrier to help-seeking was “fears of stigma and the meaning of the thought contents,”6 perhaps an understandable concern since a recent survey found that, when presented with a case vignette of an OCD sufferer with filicidal obsessions, only 62% of psychiatrists and trainees included OCD in their differential diagnosis, and, among these, 48% included involuntary psychiatric admission in their management recommendations.7
The symptoms of OCD are heterogeneous, but tend to involve obsessive themes of contamination, symmetry, superstition, inadvertently causing harm, or repugnant sexual, violent, or religious obsessions.8,9 These thoughts are distinguishable from the negative automatic thoughts involving themes of loss and failure10 that are common in depression. More than half of the general population also experiences unwanted intrusive thoughts, images, and impulses about sex, violence, or harm,10 but less persistently than and with less distress than with true OCS.
The cognitive-behavioral theory of OCS argues that depressed mood increases the frequency and intensity of intrusive thoughts11 and that inaccurate appraisals of these obsessions as being dangerous or very important lead to rituals (eg, cleaning, checking, reassurance-seeking, counting, repeating) to reduce distress. These rituals are negatively reinforced by temporary relief from the obsession, but prevent exposure to the obsessions and the opportunity to correct their misappraisals.9
There is evidence for a strong overlap between OCS and major depressive disorder (MDD),12 particularly a current episode of MDD.13 For example, Fullana et al14 analyzed data from the prospective Dunedin, New Zealand community study and found that 49% of individuals with major depression without comorbid OCD endorsed 1 or more OCS by age 32, most commonly repugnant thoughts and obsessions about inadvertently causing harm. OCS involving “thoughts of hurting the baby” are so common in postpartum depression as to be considered a symptom of the disorder.15,16 Finally, the ICD-10 clinical description for OCD17 indicates that OCS are highly prevalent in depression.
These findings—underrecognition of OCS and overlap of depression and OCS—lead to two clinically relevant questions: How common are unattended OCS in individuals seeking treatment for depression? Do unattended OCS adversely affect depression remission with medication treatment? These questions are important because they may result in millions of individuals around the world missing the chance for benefits from depression treatment.
We reasoned that a secondary analysis of the Sequenced Treatment Alternatives to Relieve Depression (STAR*D)18 trial database would provide data relevant to these research questions. We focused only on the first level of the trial, 12-week open treatment of depressed adults (who had not been treated with a selective serotonin reuptake inhibitor [SSRI] medication for the current major depressive episode) with citalopram at a dose of 20-60 mg/d, and not on later steps, which were intended to define the next best treatment if citalopram did not produce remission.18
The STAR*D trial (ClinicalTrials.gov Identifier: NCT00021528) was the largest effectiveness study of “real-world” depression ever conducted, enrolling more than 4,000 patients enrolled at 21 academic, clinic, and primary care sites across the United States, and focusing on remission of depressive symptoms as the primary endpoint. Although the STAR*D trial did not include a priori hypotheses about the impact of OCS, the study design specifically included the self-report Psychiatric Diagnostic Screening Questionnaire (PDSQ)19 at screening to assess for “concurrent psychiatric diagnoses.”20 A secondary post hoc analysis on the STAR*D dataset documented the negative impact on depression remission of such anxiety and substance abuse symptoms as measured by the PDSQ.21 However, no study to date has examined OCS because the STAR*D trial excluded primary OCD and because “the neurobiology and course of illness of obsessive-compulsive disorder distinguishes itself from other anxiety disorders”.21(p250) Hence, any OCS self-reported by participants would have been unattended to because they were (1) unrecognized, (2) judged to be secondary to depression, or (3) judged to be subclinical.
Our primary aims in this secondary analysis of this large cohort of depressed subjects in Level 1 of the STAR*D trial were (1) to estimate the occurrence of OCS in this large sample of depressed outpatients and (2) to determine the impact of unattended OCS on depression remission with citalopram.

- Obsessive-compulsive symptoms are frequently present in depressed outpatients, yet are often not attended to because sufferers are often reluctant to disclose these often embarrassing symptoms.
- Depressed patients should be asked directly about comorbid obsessive-compulsive symptoms because, if present, these symptoms can negatively impact clinical recovery from depression.
METHOD
Study Design and Patients
This study analyzed data collected as part of the STAR*D trial, which was designed to determine optimal treatment steps for outpatients with nonpsychotic MDD whose depression did not remit as a result of initial treatment with the SSRI citalopram. Complete rationale, design, and methods of the STAR*D trial have been described elsewhere.18 Briefly, this effectiveness trial attempted to maximize generalizability to “real-world” patients by enrolling more than 4,000 depressed outpatients aged 18-75 years seeking care in US outpatient general medical or psychiatric treatment settings between 2001 and 2004 who met criteria for DSM-IV MDD with an entry score of 14 or higher on the 17-item Hamilton Depression Rating Scale (HDRS-17).22 Exclusion criteria included bipolar disorder, anorexia nervosa or bulimia, psychotic symptoms or disorders, and, most relevant for the present study, a current primary diagnosis of OCD based on clinical interview (but not based upon the PDSQ or any structured diagnostic interview).
This report focused on baseline rating scales collected at entry for 3,984 subjects who had data for the variables of interest at entry into Level 1 of the STAR*D trial, in which all subjects underwent up to a 12-week open trial of citalopram at a dose between 20 and 60 mg/d.18 We did not focus on later steps in STAR*D, which focused on medication nonremitters in Level 1.
All procedures were approved by the institutional review board, and all risks, benefits, and adverse events associated with each treatment within the randomized treatments were explained to study participants, who were required to provide written informed consent prior to study participation.
Procedures
We obtained the STAR*D Level 1 data files from the coordinating site and converted them into STATA and SPSS formats for our statistical analyses. To capture subjects’ self-report of any OCS at study entry, our primary interest was in the obsessive-compulsive subscale of the PDSQ19,20 completed at study entry of the STAR*D trial to screen for concurrent psychiatric disorder symptoms. The Ns in this report may differ from those in other STAR*D outcome studies because they include all subjects who completed the relevant rating scales at entry into the study.
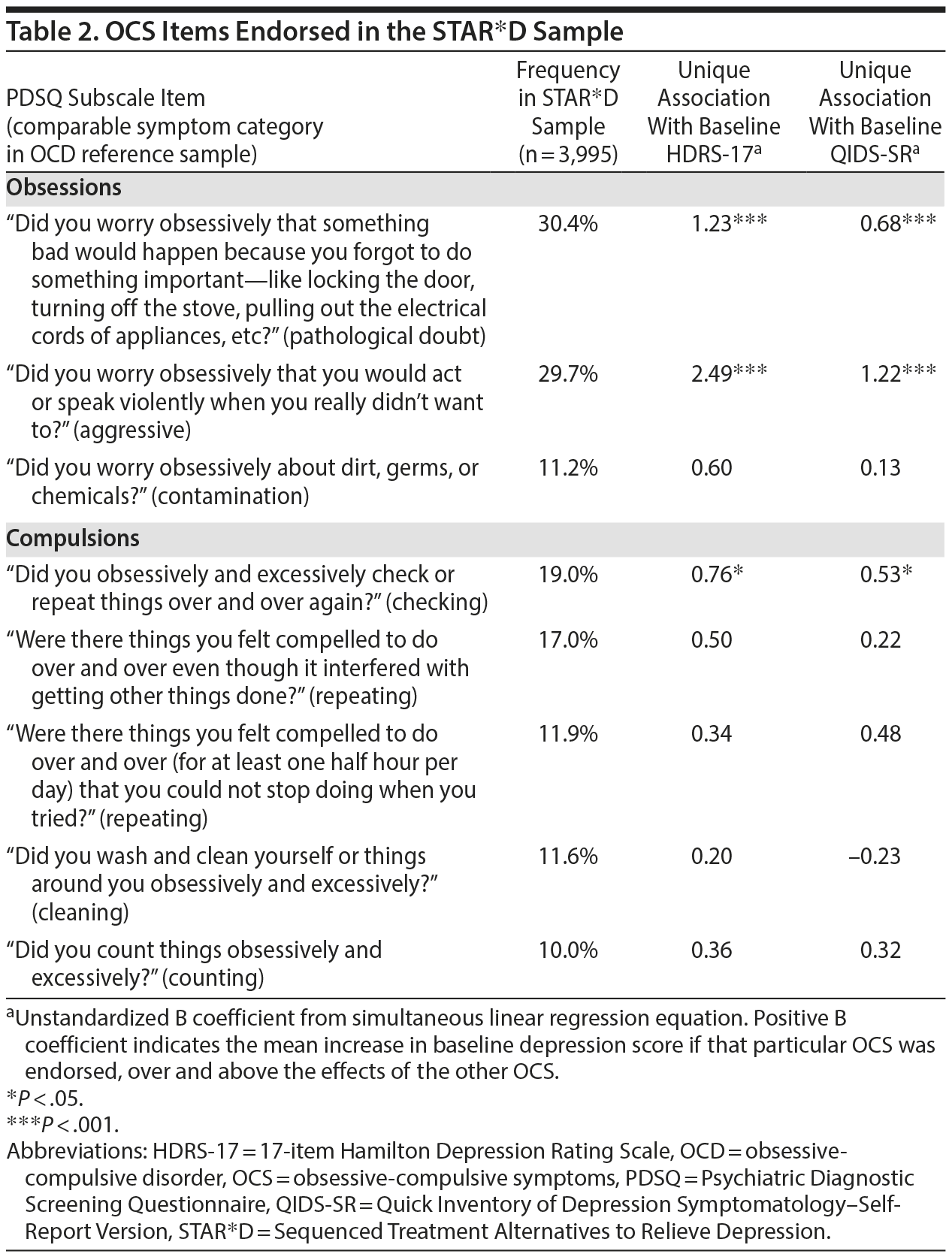
The full PDSQ consists of 139 yes/no self-report questions divided into subscales for each of the major DSM-IV Axis I psychiatric diagnoses. Our primary interest was in the 8 items constituting the obsessive-compulsive subscale of the PDSQ. These items ask the patient to answer whether he or she has experienced problems during the past 2 weeks in the following areas: (1) worrying obsessively about dirt, germs, or chemicals; (2) worrying obsessively that something bad would happen because of forgetting to do something important (eg, locking a door, turning off a stove, pulling out electrical plugs); (3) worrying obsessively about acting or speaking violently when you really didn’ t want to; (4) feeling compelled to do certain things over and over (for at least half an hour per day) that you could not stop doing when you tried; (5) feeling compelled to do things over and over even though it interfered with getting other things done; (6) obsessively and excessively washing and cleaning yourself or things around you; (7) obsessively and excessively checking or repeating things over and over again; (8) counting things obsessively and excessively.
Like the other PDSQ subscales, the OCD scale has been demonstrated to have good test retest-reliability (r > 0.80) and criterion validity compared to the Structured Clinical Interview for DSM Disorders (SCID) diagnostic interview.19 The OCD subscale has a cut-point of 1 obsessive-compulsive symptom endorsed for a sensitivity of 0.90 compared to SCID OCD diagnosis and a cut-point of 4 or more OCS endorsed for specificity of 0.90 compared to SCID OCD diagnoses.19 We chose these 2 cut-points to indicate mild and more severe degrees of OCS. Because OCD was an exclusion criterion, we did not consider these cut-points to represent an OCD diagnosis, but rather varying levels of OCS severity.
To compare the association between OCS and various severities of depression at entry into the study, we used the entry HDRS-1723 cut-points of 17 for the comparison of mild versus moderate depression and 24 for the comparison of moderate versus severe depression found optimal to maximize sensitivity and specificity.21
Following the methods of the STAR*D trial primary analyses of Level 1, we determined depression remission rates in 2 ways: (1) final HDRS-17 score of 7 or less with subjects dropped out before the final visit of Level 1 coded as “non-remitted”18 and (2) Quick Inventory of Depressive Symptomatology-Self-Report (QIDS-SR)24 final score of 5 or less, using the method of last observation carried forward.18 We examined depression remission only at the end of Level 1 because thereafter patients were assigned to various second treatments of medication or cognitive-behavioral therapy, decreasing the sample sizes and increasing the complexity of the analyses.
Statistical Analyses
Statistical analyses were conducted with the Statistical Package for the Social Sciences (SPSS) Version 20 (IBM Corporation, Armonk, New York) and included descriptive means and standard deviations for continuous variables and frequencies for categorical variables. Psychometric analyses included principal components analysis, Cronbach coefficient α, and Wilcoxon signed rank test. Inferential statistics included t tests for comparison of means and contingency analysis by χ2 for comparison of frequencies. Tests of odds ratios for depression remission were conducted using binary logistic regression. Linear regression and correlation was used with continuous and dichotomous variables. Because of the very large sample size, 2-tailed P values of .01 were considered significant, and effect sizes are presented.
RESULTS
Reliability and Validity of PDSQ OCD Subscale
Internal consistency of the 8 items of the PDSQ OCD subscale was adequate as assessed by Cronbach coefficient α = .78. The factor structure found by Zimmerman and Mattia19 for the 13 PDSQ subscales was reproduced by using principal components analysis and retaining 13 factors with varimax rotation, yielding simple structure for the 8 OCD subscale items, each of which had loadings of > 0.40 on the OCD factor and no loadings > 0.20 on any other factor. We were unable to assess test-retest reliability because the PDSQ was administered only at the screen visit. To assess discriminant validity of the PDSQ subscale, we compared the mean and median scores on this 8-item subscale to the median score on the 5-item psychotic experiences (PE) subscale, since, as with primary OCD, psychotic symptoms or disorders were excluded from the STAR*D trial.18 The means for the OCD and PE subscales were 1.41 and 0.52, respectively (dependent t3,992 = 34.17, P < .0001). The medians for the OCD and PE subscales were 1.0 and 0.0, respectively (P < .0001 by Wilcoxon signed rank test).
Frequency of OCS
Our first research question concerned the frequency of OCS in this sample of depressed patients without obvious OCD. Figure 1 demonstrates that more than half of the entire sample endorsed at least 1 OC symptom, and 14% of patients endorsed at least 4 OCS. Figure 1 also illustrates that the proportion of patients endorsing both 1 or more and 4 or more OCS at baseline increased with increasing levels of baseline depression (χ2 for each obsessive-compulsive symptom by depression level: P < .001). The linear-by-linear association between the baseline HDRS-17 depression score and number of OCS endorsed was significant (χ21 = 244.88, P < .0001; r = 0.26, P < .0001), suggesting a dose-response relationship between baseline depression and OCS frequency in this sample.
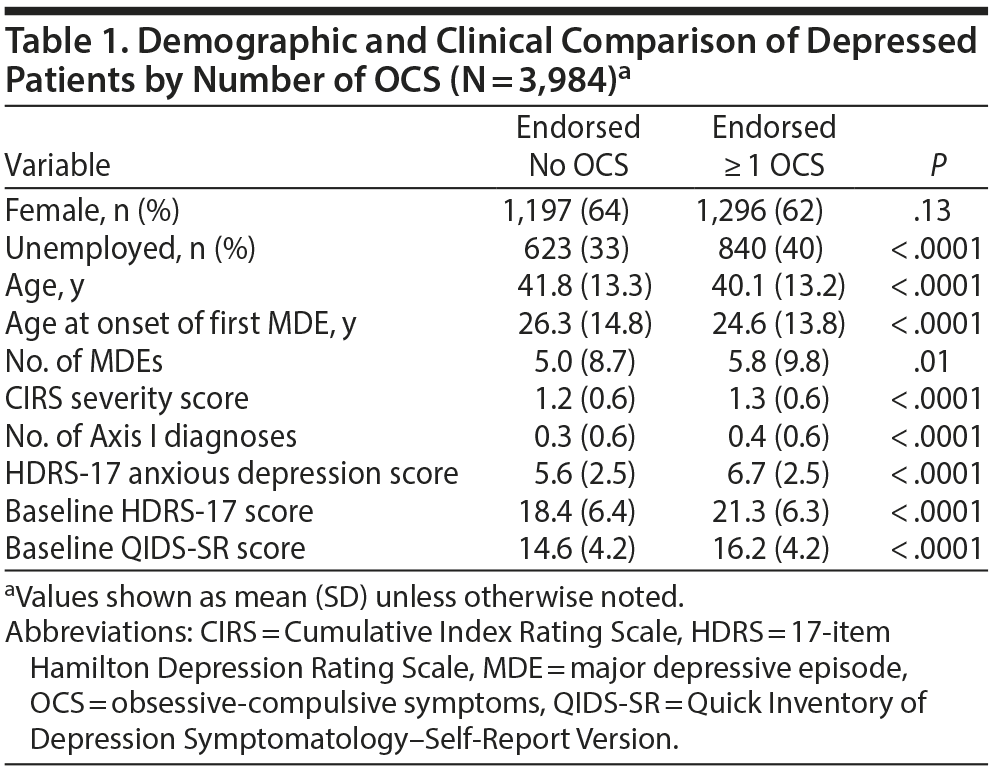
Table 1 presents a demographic and clinical comparison of depressed outpatients endorsing none versus 1 or more OCS on the PDSQ OCD subscale on entry into the STAR*D trial.
Compared to patients without OCS, patients endorsing 1 or more obsessive-compulsive symptom were significantly more likely (1) to be unemployed, (2) to be younger, (3) to have more severe MDD (as indicated by significantly earlier age at onset of first major depressive episode [MDE], more lifetime MDEs, and higher HDRS-17 and QIDS-SR scores), (4) to have more psychiatric comorbidity as indicated by the number of Axis I diagnoses based on the PDSQ, (5) to have more symptoms of anxious depression as measured by the HDRS-17, and (6) to have more physical illnesses as assessed by the Cumulative Index Rating Scale (CIRS) severity score.25 To correct for these differences, these variables and gender were included as covariates in all adjusted logistic and linear regression models.
Since OCS are often heterogeneous both between and within patients, Table 2 illustrates the relative frequencies of the various OCS probed as part of the PDSQ OCD subscale and each obsessive-compulsive symptom’s unique association with the 2 depression rating scales used in STAR*D, based on simultaneous linear regression models. As Table 2 indicates, only the 2 OCS related to harming obsessions contributed significant unique predictive value over and above the other OCS at P < .001.
When all 8 OCS were simultaneously entered into separate simultaneous ordinal regression equations (using the logit link function) to predict the HDRS-17 and QIDS-SR suicide item scores, only violent obsessions contributed significant, unique variance to the prediction (both P < .001).
Impact of OCS on Depression Remission
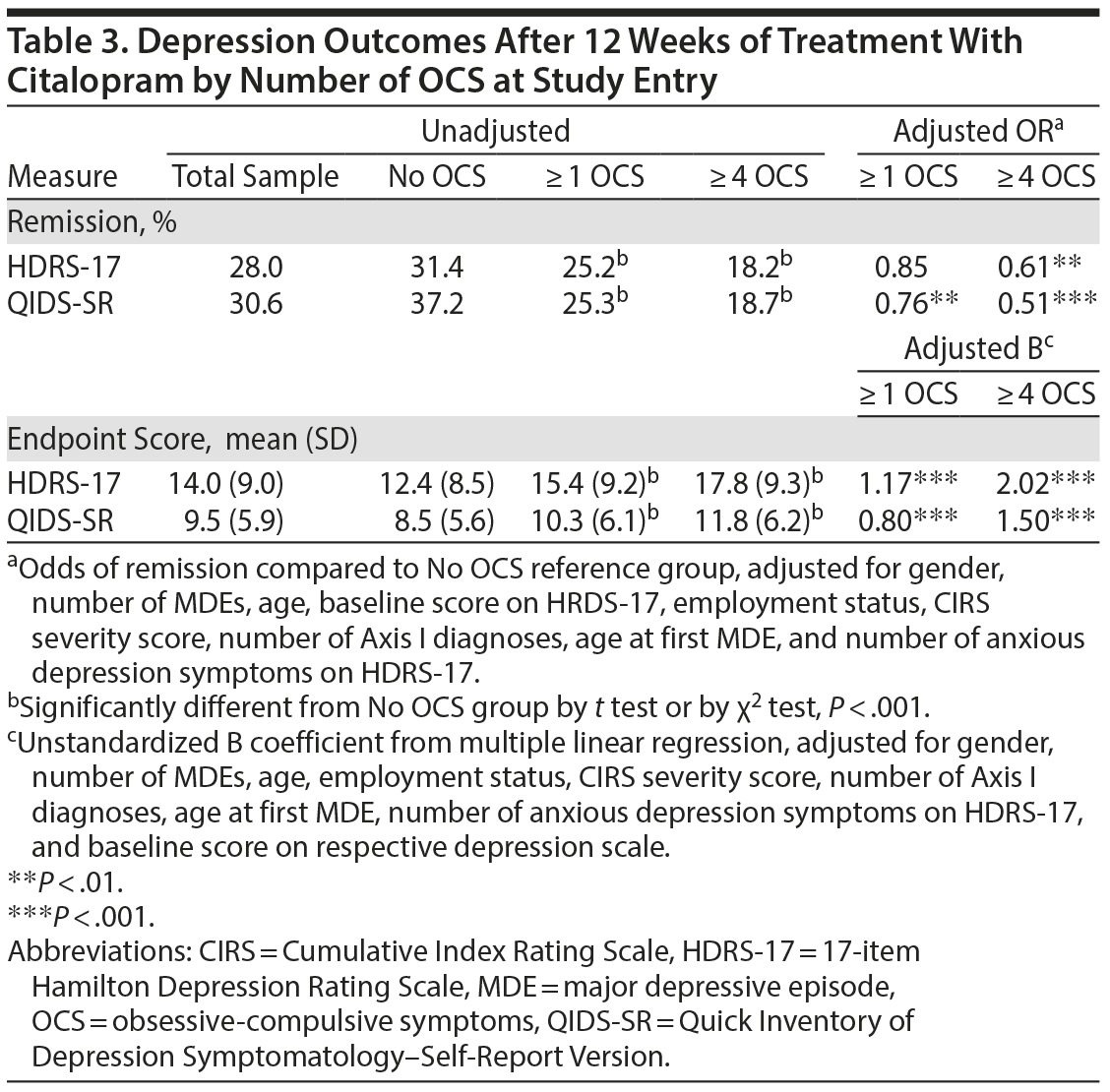
Our second research question was, if a significant percentage of STAR*D patients had OCS, would this make a practical difference in the effects of their antidepressant medication treatment for depression? Table 3 illustrates that while the sample as a whole remitted at a 28% rate by HDRS-17 criteria at 12 weeks, the remission rate for patients with no OCS was 31%, and remission rates were significantly lower for patients endorsing 1 or more (25%) or 4 or more OCS (18%); a similar pattern was seen on remission as defined by self-reported QIDS score. In all cases, raw remission rates for subjects endorsing 1 or more OCS or ≥ 4 OCS were significantly lower than subjects endorsing no OCS (all P < .0001). Table 3 also contains adjusted odds ratios from logistic regression models of remission and unstandardized B weights from linear regression models treating final depression level as a dimensional or continuous variable. These indicate that with correction, all remission and dimensional differences survived correction at P < .01 with the exception of subjects endorsing 1 or more OCS and remission on HDRS-17.
DISCUSSION
Our results indicate that OCS are highly prevalent in depressed outpatients and are commonly not attended to. In a representative sample of nonpsychotic depressed outpatients that excluded primary cases of OCD, the majority endorsed at least 1 obsessive-compulsive symptom—most commonly obsessions about harm, which mirrored the results of the Dunedin community study.14 Harming obsessions were found to be uniquely related to depression severity in the present study, which is consistent with previous research indicating that they severely impact sufferers’ quality of life.26
OCS impacted depression remission and depression improvement as measured by both clinician- and patient-rated measures. A greater number of OCS was associated with lower odds of depression remission. We also observed a close relation between OCS and severity of depression, with frequency of endorsement of all symptoms increasing along with patients’ depression level. In addition, presence of OCS was related to more severe depression as indicated by earlier onset of first MDE and more total lifetime MDEs. This finding is consistent with previous research indicating greater impairment in patients with comorbid MDD and OCD.27
Our findings are correlational in nature and cannot indicate direction of causation between depression and OCS, and it is likely that there is a feedback loop between these symptoms. Both depression and obsessions are believed to be linked to dysregulation in the brain’s serotonin system,9,12 and a recent large twin study found that OCS and depressive symptoms may occur together primarily as a result of shared genetic factors.28
Limitations of our secondary analysis of the existing STAR*D dataset include the absence of a psychometric instrument specifically designed to assess OCS, such as the Yale-Brown Obsessive Compulsive Scale and Symptom Checklist29 (although the PDSQ has demonstrated good criterion validity with a SCID diagnosis of OCD, it has not yet been compared statistically to these other validated measures of obsessive-compulsive severity). In addition, OCS were assessed only at study entry, which prohibited assessment of their change with treatment. Finally, exclusion of patients with primary OCD from the STAR*D trial makes it impossible for us to comment on the prevalence of underrecognized OCD diagnoses in this study. Future studies should address these limitations.
CONCLUSION
OCS are common in depressed outpatients and are often not attended to. They impact clinical recovery from depression and should be screened for, since sufferers are often reluctant to disclose these symptoms. We would expect that OCS are likely to be more common in depressed outpatients, since the STAR*D sample excluded subjects with primary OCD.
Not attending to OCS may result in patients’ missing the chance for successful treatment of both their depressive and their obsessive-compulsive symptoms (since treatment of OCD and major depression differ in that [1] specific cognitive-behavioral techniques are generally required for OCS9 and [2] patients with OCS may require a higher dose of antidepressants than patients with major depression).30 These findings highlight the need to improve our training of psychiatric and primary care clinicians to recognize and effectively treat depressed patients with OCS.
The relatively high frequency of OCS present in this sample suggests that screening for OCS in depressed outpatients with questions similar to those in Table 2 would be efficient and would help reduce our patients’ suffering. Particularly important OCS symptoms to screen for are harming obsessions, which were the most common in our sample and yet also the symptoms least commonly shared with treaters due to stigma.6
Submitted: November 19, 2014; accepted March 12, 2015.
Drug name: citalopram (Celexa and others).
Disclosure of off-label usage: The authors have determined that, to the best of their knowledge, no investigational information about pharmaceutical agents that is outside US Food and Drug Administration-approved labeling has been presented in this activity.
Author contributions: All authors had access to all the data in the study and had final responsibility for the decision to submit for publication.
Financial disclosures: Dr Trivedi is or has been an advisor/consultant to Abbott, Abdi Ibrahim, Akzo (Organon), Alkermes, AstraZeneca, Axon Advisors, Bristol-Myers Squibb Company, Cephalon, Cerecor, Concert, Eli Lilly, Evotec, Fabre-Kramer, Forest, GlaxoSmithKline, Janssen, Johnson & Johnson, Libby, Lundbeck, Meade Johnson, MedAvante, Medtronic, Merck, Mitsubishi Tanabe, Naurex, Neuronetics, Otsuka, Pamlab, Parke-Davis, Pfizer, Pgx Health, Phoenix Marketing Solutions, Rexahn, Ridge Diagnostics, Roche, Sepracor, Shire, Sierra, SK Life and Science, Sunovion, Takeda, Tal Medical/Puretech, Targacept, Transcept, VantagePoint, Vivus, and Wyeth-Ayerst and has received research support from Agency for Healthcare Research and Quality, Corcept, Cyberonics, National Alliance for Research on Schizophrenia and Depression (NARSAD), National Institute of Mental Health (NIMH), and National Institute on Drug Abuse (NIDA). Dr Rush reports personal fees from the American Psychiatric Association, NIDA, Eli Lilly, Lundbeck, Medavante, Guilford Press,Penn State Hershey Medical Center, American Society of Clinical Psychopharmacology (ASCP), University of California at San Diego, UT Southwestern Medical Center Dallas, Santium, Stanford University, and Duke-NUS; grants from Duke-NUS; and non-financial support from the International College of Neuropsychopharmacology. Dr Fava has received research support from Abbott; Alkermes; American Cyanamid; Aspect Medical Systems; AstraZeneca; Avanir; BioResearch; BrainCells; BristolMyers Squibb; CeNeRx BioPharma; Cephalon; Clintara; Covance; Covidien; Eli Lilly; EnVivo; Euthymics Bioscience; Forest; Ganeden Biotech; GlaxoSmithKline; Harvard Clinical Research Institute; Hoffman-LaRoche; Icon Clinical Research; i3 Innovus/Ingenix; Janssen; Jed Foundation; Johnson & Johnson; Lichtwer; Lorex; Lundbeck; MedAvante; Methylation Sciences; NARSAD; National Center for Complementary and Alternative Medicine; NIDA; NIMH; Neuralstem; Novartis; Organon; Pamlab; Pfizer; Pharmacia & Upjohn; Pharmaceutical Research Associates; Pharmavite; Pharma-Rx; Photothera; Reckitt Benckiser; Roche; RCT Logic; SanofiAventis; Shire; Solvay; Stanley Medical Research Institute; Synthelabo; and Wyeth-Ayerst; has been an advisor/consultant for Abbott; Affectis; Alkermes; Amarin; Aspect Medical Systems; AstraZeneca; Auspex; AvanirAxsomeBayer; Best Practice Project Management; BioMarin; Biovail; BrainCells; BristolMyers Squibb; CeNeRx BioPharma; Cephalon; Cerecor; CNS Response; Compellis; Cypress; DiagnoSearch Life Sciences; Dainippon Sumitomo; Dov; Edgemont; Eisai; Eli Lilly; EnVivo; ePharmaSolutions; Epix; Euthymics Bioscience; Fabre-Kramer; Forest; Genomind; GlaxoSmithKline; Grunenthal; i3 Innovus/Ingenix; Janssen; Jazz; Johnson & Johnson; Knoll; Labopharm; Lorex; Lundbeck; MedAvante; Merck; MSI Methylation Sciences; Naurex; Neuralstem; Neuronetics; NextWave; Novartis; Nutrition 21; Orexigen; Organon; Otsuka; Pamlab; Pfizer; PharmaStar; Pharmavite; Pharma-Rx; Precision Human Biolaboratory; Prexa; Puretech; PsychoGenics; Psylin Neurosciences; RCT Logic; Rexahn; Ridge Diagnostics; Roche; SanofiAventis; Sepracor; Servier; Schering-Plough; Solvay; Somaxon; Somerset; Sunovion; Supernus; Synthelabo; Takeda; Tal Medical; Tetragenex; TransForm; Transcept; and Vanda; has received speaking/publishing fees from Adamed; Advanced Meeting Partners; American Psychiatric Association; ASCP; AstraZeneca; Belvoir Media Group; Boehringer Ingelheim; Bristol-Myers Squibb; Cephalon; Physicians Postgraduate Press; Eli Lilly; Forest; GlaxoSmithKline; Imedex; Massachusetts General Hospital (MGH) Psychiatry Academy/Primedia; MGH Psychiatry Academy/Reed Elsevier; Novartis; Organon; Pfizer; PharmaStar; United BioSource; and Wyeth-Ayerst; has equity holdings in Compellis and PsyBrain; holds a patent for Sequential Parallel Comparison Design, licensed by MGH to Pharmaceutical Product Development, and a patent application for A Combination of Ketamine Plus Scopolamine in Major Depressive Disorder; holds copyrights for the MGH Cognitive & Physical Functioning Questionnaire, the Sexual Functioning Inventory, the Antidepressant Treatment Response Questionnaire, the Discontinuation-Emergent Signs & Symptoms checklist, and the SAFER interview; and receives royalties from Lippincott, Williams & Wilkins; Wolters Kluwer; and World Scientific Publishing. Drs Baer and Wisniewski and Ms Huz have no personal affiliations or financial relationships with any commercial interest to disclose relative to the article.
Funding/support: Chirag Foundation.
Role of the sponsor: The funder of this study had no role in the study design, data collection, data analysis, data interpretation, or writing of this report.
Acknowledgments: Data used in the preparation of this article were obtained from the limited access data sets distributed from the NIH-supported STAR*D. Sequenced Treatment Alternatives to Relieve Depression focused on nonpsychotic major depressive disorder in adults seen in outpatient settings. The primary purpose of this research study was to determine which treatments worked best if the first treatment with medication does not produce an acceptable response. The study was supported by the National Institute of Mental Health (NIMH) Contract # N01MH90003 to the University of Texas Southwestern Medical Center.
Additional information: Requests for access to the STAR*D data set are made to the NIMH, sponsor of the trial. See http://www.nimh.nih.gov/trials/data sets/nimh-procedures-for-requesting-data-sets.shtml for information.
Find more articles on this and other psychiatry and CNS topics:
The Journal of Clinical Psychiatry
The Primary Care Companion for CNS Disorders
REFERENCES
1. Kessler RC, Chiu WT, Demler O, et al. Prevalence, severity, and comorbidity of 12-month DSM-IV disorders in the National Comorbidity Survey Replication. Arch Gen Psychiatry. 2005;62(6):617-627. PubMed doi:10.1001/archpsyc.62.6.617
2. Subramaniam M, Abdin E, Vaingankar JA, et al. Obsessive-compulsive disorder: prevalence, correlates, help-seeking and quality of life in a multiracial Asian population. Soc Psychiatry Psychiatr Epidemiol. 2012;47(12):2035-2043. PubMed doi:10.1007/s00127-012-0507-8
3. Gururaj GP, Math SB, Reddy JYC, et al. Family burden, quality of life and disability in obsessive compulsive disorder: an Indian perspective. J Postgrad Med. 2008;54(2):91-97. PubMed doi:10.4103/0022-3859.40773
4. Wahl K, Kordon A, Kuelz KA, et al. Obsessive-Compulsive Disorder (OCD) is still an unrecognised disorder: a study on the recognition of OCD in psychiatric outpatients. Eur Psychiatry. 2010;25(7):374-377. PubMed doi:10.1016/j.eurpsy.2009.12.003
5. Glazier K, Calixte RM, Rothschild R, et al. High rates of OCD symptom misidentification by mental health professionals. Ann Clin Psychiatry. 2013;25(3):201-209. PubMed
6. Belloch A, Del Valle G, Morillo C, et al. To seek advice or not to seek advice about the problem: the help-seeking dilemma for obsessive-compulsive disorder. Soc Psychiatry Psychiatr Epidemiol. 2009;44(4):257-264. PubMed doi:10.1007/s00127-008-0423-0
7. Booth BD, Friedman SH, Curry S, et al. Obsessions of child murder: underrecognized manifestations of obsessive-compulsive disorder. J Am Acad Psychiatry Law. 2014;42(1):66-74. PubMed
8. Pinto A, Mancebo MC, Eisen JL, et al. The Brown Longitudinal Obsessive Compulsive Study: clinical features and symptoms of the sample at intake. J Clin Psychiatry. 2006;67(5):703-711. PubMed doi:10.4088/JCP.v67n0503
9. Abramowitz JS, Taylor S, McKay D. Obsessive-compulsive disorder. Lancet. 2009;374(9688):491-499. PubMed doi:10.1016/S0140-6736(09)60240-3
10. Purdon C, Clark DA. Obsessive intrusive thoughts in nonclinical subjects, part 1: content and relation with depressive, anxious and obsessional symptoms. Behav Res Ther. 1993;31(8):713-720. PubMed doi:10.1016/0005-7967(93)90001-B
11. Rachman S. A cognitive theory of obsessions. Behav Res Ther. 1997;35(9):793-802. PubMed doi:10.1016/S0005-7967(97)00040-5
12. Murphy DL, Moya PR, Fox MA, et al. Anxiety and affective disorder comorbidity related to serotonin and other neurotransmitter systems: obsessive-compulsive disorder as an example of overlapping clinical and genetic heterogeneity. Philos Trans R Soc Lond B Biol Sci. 2013;368(1615):20120435. PubMed
13. Thaipisuttikul P, Ittasakul P, Waleeprakhon P, et al. Psychiatric comorbidities in patients with major depressive disorder. Neuropsychiatr Dis Treat. 2014;10:2097-2103. PubMed
14. Fullana MA, Mataix-Cols D, Caspi A, et al. Obsessions and compulsions in the community: prevalence, interference, help-seeking, developmental stability, and co-occurring psychiatric conditions. Am J Psychiatry. 2009;166(3):329-336. PubMed doi:10.1176/appi.ajp.2008.08071006
15. Depression during and after pregnancy, fact sheet 2009. Office on Women’s Health, US Department of Health and Human Services Web site. https://www.womenshealth.gov/publications/our-publications/fact-sheet/depression-pregnancy.html. Updated July 16, 2012. Accessed August 24, 2015.
16. Wisner KL, Peindl KS, Gigliotti T, et al. Obsessions and compulsions in women with postpartum depression. J Clin Psychiatry. 1999;60(3):176-180. PubMed doi:10.4088/JCP.v60n0305
17. The ICD-10 Classification of Mental and Behavioral Disorders: clinical descriptions and diagnostic guidelines, pp 117. World Health Organization Web site. http://www.who.int/classifications/icd/en/bluebook.pdf. Accessed July 23, 2015.
18. Rush AJ, Fava M, Wisniewski SR, et al; STAR*D Investigators Group. Sequenced Treatment Alternatives to Relieve Depression (STAR*D): rationale and design. Control Clin Trials. 2004;25(1):119-142. PubMed doi:10.1016/S0197-2456(03)00112-0
19. Zimmerman M, Mattia JI. A self-report scale to help make psychiatric diagnoses: the Psychiatric Diagnostic Screening Questionnaire. Arch Gen Psychiatry. 2001;58(8):787-794. PubMed doi:10.1001/archpsyc.58.8.787
20. Zimmerman M. The Psychiatric Diagnostic Screening Questionnaire. Los Angeles, CA: Western Psychological Services; 2002.
21. Howland RH, Rush AJ, Wisniewski SR, et al. Concurrent anxiety and substance use disorders among outpatients with major depression: clinical features and effect on treatment outcome. Drug Alcohol Depend. 2009;99(1-3):248-260. PubMed doi:10.1016/j.drugalcdep.2008.08.010
22. Hamilton M. Rating depressive patients. J Clin Psychiatry. 1980;41(12 pt 2):21-24. PubMed
23. Zimmerman M, Martinez JH, Young D, et al. Severity classification on the Hamilton Depression Rating Scale. J Affect Disord. 2013;150(2):384-388. PubMed doi:10.1016/j.jad.2013.04.028
24. Rush AJ, Trivedi MH, Ibrahim HM, et al. The 16-Item Quick Inventory of Depressive Symptomatology (QIDS), clinician rating (QIDS-C), and self-report (QIDS-SR): a psychometric evaluation in patients with chronic major depression. Biol Psychiatry. 2003;54(5):573-583. PubMed doi:10.1016/S0006-3223(02)01866-8
25. Hudon C, Fortin M, Vanasse A. Cumulative Illness Rating Scale was a reliable and valid index in a family practice context. J Clin Epidemiol. 2005;58(6):603-608. PubMed doi:10.1016/j.jclinepi.2004.10.017
26. Brakoulias V, Starcevic V, Berle D, et al. The characteristics of unacceptable/taboo thoughts in obsessive-compulsive disorder. Compr Psychiatry. 2013;54(7):750-757. PubMed doi:10.1016/j.comppsych.2013.02.005
27. Hofmeijer-Sevink MK, van Oppen P, van Megen HJ, et al. Clinical relevance of comorbidity in obsessive compulsive disorder: the Netherlands OCD Association study. J Affect Disord. 2013;150(3):847-854. PubMed doi:10.1016/j.jad.2013.03.014
28. Kendler KS, Campbell J. Expanding the domain of the understandable in psychiatric illness: an updating of the Jasperian framework of explanation and understanding. Psychol Med. 2014;44(1):1-11. PubMed doi:10.1017/S0033291713001591
29. Goodman WK, Price LH, Rasmussen SA, et al. The Yale-Brown Obsessive Compulsive Scale, 1: development, use, and reliability. Arch Gen Psychiatry. 1989;46(11):1006-1011. PubMed doi:10.1001/archpsyc.1989.01810110048007
30. Shim G, Park HY, Jang JH, et al. What is the optimal dose of escitalopram for the treatment of obsessive-compulsive disorder? a naturalistic open-label study. Int Clin Psychopharmacol. 2011;26(5):284-290. PubMed doi:10.1097/YIC.0b013e32834a5c09
Save
Cite
Advertisement
GAM ID: sidebar-top